TOUR OF THE INFINITELY TINY:
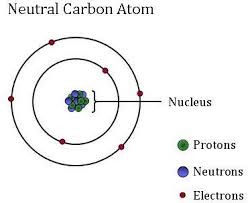
At the base of all of the immense grandeur of the universe, are atoms made up of negatively charged Electrons orbiting nuclei composed of positively charged Protons and electrically neutral Neutrons. The atom is the smallest unit that still can be identified as a particular chemical element. If that was the whole picture, it would be relatively simple. However, there are even finer divisions and many more particles. Let me see if I can give you an overview without getting too deep or confusing. I apologize in advance for any tedious or confusing details. If you want more details, check it out in popular textbooks.
According to the Standard Model of particle physics, Protons and Neutrons are actually composed of three smaller entities called Quarks and are thus classified as Baryons, while Electrons are indivisible and are classified as Leptons. The Neutron consists of two down and one up Quark held together by Gluons, (a particle that carries force); the Proton consists of two up and one down Quark also held together by Gluons. There are actually six types of Quarks with the frivolous names of up, down, top, bottom, charm and strange[1], and six types of Leptons called Electron, Muon, Tau, Electron Neutrino, Muon Neutrino and Tau Neutrino particles.
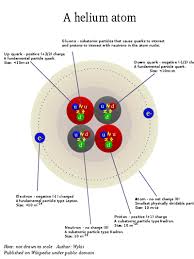 There are also anti-particles for each of these, such as the Positron, the antiparticle to the Electron, Antiproton, Antineutron, Muon Antineutrino, etc. Matter and Antimatter, when combined, annihilate each other with the release of tremendous energy. Einstein’s formula, E=MC2, means the energy you can get from matter is the mass times the speed of light squared. That’s a lot of energy for a tiny bit of mass and is the basis of the power of the atomic bomb, which splits atoms to release energy. It is also the basis of fusion energy in the sun which combines four hydrogen atoms in a three step process to form a helium atom with the release of essentially mass-less Electron Neutrinos and energy in the form of gamma rays from the matter-antimatter annihilation of a released positron by an electron. These gamma rays work their way to the sun’s surface through countless collisions, each of which reduces their energy and frequency, transforming them into various frequencies of light, e.g. ultraviolet, visible and infrared.
There are also anti-particles for each of these, such as the Positron, the antiparticle to the Electron, Antiproton, Antineutron, Muon Antineutrino, etc. Matter and Antimatter, when combined, annihilate each other with the release of tremendous energy. Einstein’s formula, E=MC2, means the energy you can get from matter is the mass times the speed of light squared. That’s a lot of energy for a tiny bit of mass and is the basis of the power of the atomic bomb, which splits atoms to release energy. It is also the basis of fusion energy in the sun which combines four hydrogen atoms in a three step process to form a helium atom with the release of essentially mass-less Electron Neutrinos and energy in the form of gamma rays from the matter-antimatter annihilation of a released positron by an electron. These gamma rays work their way to the sun’s surface through countless collisions, each of which reduces their energy and frequency, transforming them into various frequencies of light, e.g. ultraviolet, visible and infrared.
In addition to these particles, there are particles that carry the forces that maintain and govern the behavior of atoms. There are three gauge Bosons (called W Boson, Z Boson, Gluon) and the Photon, as well as the Higgs Boson that may have been found but has not been confirmed experimentally. The W and Z Bosons carry the nuclear Strong and Weak Forces holding the nucleus together and responsible for radioactive decay respectively. The Gluons (of which there are 8 varieties) hold the Quarks of Neutrons and Protons together as noted earlier. The Photon carries the Electromagnetic Force that holds the electrons to the nucleus.
The Higgs Boson is theorized to be responsible for mass or gravity and is necessary because quantum theory assumes massless particles. If found it could, potentially, lead to a “Theory of Everything” which includes the fourth force, Gravity. At present, the picture of the Weak Nuclear, Strong Nuclear and Electromagnetic forces, defined by the Standard Model of quantum physics, is incompatible with our picture of Gravity by Einstein’s Theory of Relativity. One or both of these theories may be wrong or at least incomplete.
An electron can be bumped to a higher energy orbital within an atom by absorbing energy and then it will fall back to a more stable, lower-energy orbital by emitting the energy as a photon. Only photons of a specific energy (frequency) can be absorbed by any one electron depending on its energy level. These distinct energies are called quanta as are the distinct orbital levels, thus the name “quantum physics.” There is always a loss of energy in this process, so that the photon that is emitted when the electron falls back to its ground state is always at a lower energy than the photon that is absorbed. The absorption or emission of photons at distinct frequencies gives us a powerful tool to identify what elements are present. The family of absorption or emission lines in the spectra of stars is based on this principle and is used to identify their elemental content.
The mass of the Neutron is 1.6749×10−24 grams. The mass of the Proton is 1.6726×10−24 grams. The mass of the Electron is 9.1094×10−27 grams. More simply, the Electron is 1/1836th of the mass of the Proton or 1/1839th of the Neutron, so the atomic weight of an element is mostly due to the Protons and Neutrons in the nucleus. Sorry, there is no easy way to illustrate these tiny weights, but maybe it will help to put them in more common terms. To make one ounce, it would take 1.6926x 1025 Neutrons, or 1.6949x 1025 Protons or 3.1121 x 1027 Electrons[2]. The elements from which all matter is composed consist of atoms with varying numbers of neutrons, protons and electrons, usually in equal numbers except for isotopes having one or more neutrons less or more.
There are 94 naturally occurring elements and 24 confirmed heavier elements created artificially. The lightest element is Hydrogen with only one electron encircling one Proton (atomic weight 1.00794 g/mole), then Helium with two Electrons encircling a nucleus of two Protons and two Neutrons (atomic weight 4.002602 g/mole). Some important elements include carbon with 6 Electrons, Protons and Neutrons (atomic weight 12.0107 g/mole), nitrogen at 7 of each (atomic weight 14.0067 g/mole), and oxygen, at 8 of each (atomic weight 15.9994 g/mole). Plutonium-94, atomic weight approximately 242, is the heaviest confirmed naturally occurring element, and Ununoctium-118, approximate atomic weight 294, is the heaviest confirmed synthetic element to date (ca. 2010).
A mole of a substance has mass in grams equal to the substance’s molecular or atomic weight[3]. Most elements also have isotopes, usually containing added or reduced numbers of neutrons, naturally occurring as a small percentage of the total. Note that atomic weights listed are actually the averages of all isotopes in their naturally occurring ratios. Some of these isotopes are unstable and decay to the stable state radioactively at a specific unique rate, i.e. half-life. One good example is Carbon, with a standard atomic weight of approximately 12, but that can occur as Carbon-13 and Carbon-14, containing one and two extra Neutrons respectively.
 Solar Fusion of Hydrogen into Helium-4
Solar Fusion of Hydrogen into Helium-4
In the core of the sun the temperature is so high that protons and electrons from hydrogen are essentially free and reach speeds high enough to fuse. (Temperature is a measure of the average speed of particles, the higher the faster.)
- In step one, two protons fuse into a deuteron, made up of one proton and one neutron by converting one proton into a neutron with the release of an almost massless electron neutrino and a positron (antiparticle of the electron). In the sea of fast and free electrons, the positron quickly finds and annihilates with an electron releasing tremendous energy in the form of gamma rays.
2. In step two, another proton fuses with each deuteron to form helium-3, containing two protons and one neutron.
3. In step three, two helium-3 nuclei fuse and form a helium-4 nucleus, containing two protons and two neutrons, with the release of two protons.
The total reaction to produce helium-4 uses two electrons and six protons, releasing two protons at the end, and by converting two protons to two neutrons, releasing two positrons and two neutrinos. Additional gamma rays released by binding energy are produced with each fusion event in the chain.
The two positrons annihilate with two electrons releasing gamma rays which may take thousands of years to reach the surface of the sun through countless interactions with matter. Losing energy with each interaction, they appear mostly as UV, visible and infrared light at the surface.
The two electron neutrinos, which rarely interact with other matter, leave the sun immediately, bombarding the earth with an estimated trillion particles per meter each second. Most of these neutrinos pass harmlessly through the earth and continue on their way. The sun is estimated to convert 600 million metric tons per second of hydrogen into helium-4, annihilating 4.3 million metric tons per second into pure energy. Only about a third of the expected electron neutrinos are detected on earth, so physicists have theorized that neutrinos oscillate between the three forms, electron, muon and tau neutrinos. The other two forms require different types of detectors. So far, muon and tau neutrinos have only been detected in particle accelerator experiments, but oscillation from electron neutrino to muon neutrino may recently have been detected from space.
Carbon-14 is created when thermal neutrons, formed by cosmic rays, hit Nitrogen-14 in the atmosphere[4]. Carbon-14 decays with a half-life of 5,730 ± 40 years back into stable Nitrogen-14. The term half-life means that half of the mass is radioactively converted into its more stable form in that time. Plants take in the Carbon-14 as Carbon Dioxide and animals eat plants or other animals that ate plants, so their bodies approximate the level of Carbon-14 in the atmosphere (or ocean). Accumulation stops when they die. The convenient half-life length makes it a good marker to date ancient organic artifacts up to 60,000 years old[5]. Unlike Carbon-14, Carbon-13 is a relatively stable isotope that is present at approximately 1% in all carbon on earth. Isotopes have similar but different properties from the standard state due to the difference in weight.
The diameter of the smallest atom, Helium, enclosing the nucleus and two electrons orbiting it, is approx. 64 x 10-12 meters (64 picometers)[6]. The diameter of the Helium nucleus, containing 2 Neutrons and 2 Protons, is about 3.4 x 10-15 (3.4 femtometers). So the diameter of this atom is 18,692 times the diameter of the nucleus, or over 6.5 x 1012 (6.5 trillion) times the volume of the nucleus. This means that the Helium atom is made up of mostly empty space between the nucleus and the electrons. This is true within and between all atoms and molecules.
Space, the Final Frontier
What appears to be solid matter on the larger scale, is really atoms containing, and contained in, mostly empty space interacting with other atoms electronically in an ordered system. It is possible for a photon or other particle to pass through without ever hitting a single atom or subatomic particle. This is precisely what happens when X-Rays pass through the body, only leaving shadows on the film where some of the rays encounter denser material like bone. This is why the trillion neutrinos per meter per second, formed and emitted by the sun, can pass through the entire earth all of the time, rarely hitting any other particle.
So if the universe is composed of 700,000 trillion, trillion times as much space as matter, and even the matter, within and between atoms, is mostly empty space, it could be said that statistically the amount of matter in the universe is negligible or practically nonexistent.
Molecules – The Basics
Atoms of the elements combine into molecules in pairs, clusters, branched and unbranched chains, rings, etc. and form solids by arranging themselves in lattices (crystals like diamond), sheets (like graphite) and glasses (poorly ordered conglomerations), or as liquids and gases which are less ordered, depending on the temperature. Everything we see, feel or interact with is composed of chemicals made of elements or molecules, all made of atoms. Everything that physically exists is composed of chemicals, so, if a commercial product is advertised as “chemical free” they are lying. They may mean there are no synthetic (man-made) chemicals added, but the product cannot be chemical free if it exists.
Atmospheric gases are small molecules in which usually two or more atoms or elements combine, such as N2, O2, Ar, CO2, H2. Air contains 78% Nitrogen, 21% Oxygen, 0.9% Argon and 0.038% Carbon Dioxide, but Hydrogen is not a significant component of air unless you count water vapor, average 1%, dissolved in the air. Note that Argon (Ar) is not paired because it is a “noble gas” like helium and neon that do not combine readily because their electron orbitals are “complete” i.e. have just the right number of electrons to complete each orbital making them stable. Combine Hydrogen and Oxygen (two gases) to get H2O, water, which can be a liquid, solid or gas depending on the temperature and pressure.
When UV rays from the sun or lightning split water vapor, Hydrogen and Oxygen[7] are released as charged atoms called ions. These are very chemically active and quickly combine with other atoms to form, for example, Ozone, a very chemically unstable compound made up of three Oxygen atoms. UV rays from the sun continually produce Ozone by splitting and recombining Oxygen atoms in the upper layers of the atmosphere by this process, and the Ozone helps to shield the earth from harmful UV rays.
When rain water combines with Carbon Dioxide, Carbonic Acid is formed, (CO2 + H2O = H2CO3 or as ions HCO3– + H+). Rain water is usually slightly acidic at pH 5 or so (not neutral pH 7) because of these reactions. As a matter of fact, any water left sitting open to the air will assume a similar acidic pH by absorbing Carbon Dioxide from the air unless buffered by other molecules. Buffering is a process where two components “fight” each other to maintain a particular pH balance. Fortunately, most systems containing living organisms contain ions that are very good buffers such as Phosphates and Carbonates (PO4-3 and CO3-2). Due to this buffering effect, oceans average pH above 8 (slightly alkaline) in spite of the fact that 50% of CO2 produced is absorbed by the oceans.
The most abundant element, by weight, on earth is Oxygen at 46% of crust, 79% of ocean water, 65% of the human body and is only surpassed by Iron in the total composition of the earth at 30%, with Iron (mostly core) estimated at 35%. Living things are based on large carbon-based molecules containing oxygen, hydrogen and nitrogen with a smattering of other elements such as phosphorus, sulfur and calcium. Nonliving things are mostly composed of other elements such as silicon, magnesium, aluminum and iron, usually in complex multi-element combinations with oxygen, sulfur, etc. Nonliving (inorganic) compounds can contain carbonates, phosphates, etc. which may have been originally derived from living things (organic). For instance, limestone is composed largely of calcium and magnesium carbonates and oxides that may have originally derived from the shells and skeletons of microscopic life laid down over millennia as sediments in shallow seas.
[1] The types of Quarks have frivolous, made-up names, as does the Quark itself.
[2] Note that 1027 is a 1 with 27 zeros after it and 1025 is a 1 with 25 zeros after it, and that 1027 is 100 times 1025.
[3] This is a simplification but in essence is true. It is based on Avogadro’s number, 6.0221 x 1023 which is the number of atoms contained in one gram-molecular weight or mole of an element.
[4] Carbon-14 also occurs in fossil fuels and crystalline minerals such as diamond. The source is uncertain, but radioactive elements may create it continually, such as when radioactive Radium-223 decays to form Carbon-14 and Lead- 209.
[5] Approximately 10.5 half lives. Each half life halves the amount of carbon -14 remaining, so that would result in approximately 9.7 x 10-4 of the original amount of carbon-14 remaining.
[6] Helium is smaller in size, but approximately four times the mass of Hydrogen. Its smaller size is due to the electrons being more tightly held by the two protons in the nucleus than those of Hydrogen which has only one proton. Additionally, Hydrogen is usually found as a molecule composed of two Hydrogen atoms (H2) or combined with other elements.
[7] Simplified version, since hydroxyl ions (OH–) are also produced.