science and philosophy
RuvAB: Another Elegant Molecular Motor Visualized — Evolution News
This machine looks like something out of a CAD/CAM project, but it’s found in bacteria. Source
RuvAB: Another Elegant Molecular Motor Visualized — Evolution News
Climate Change: What’s Real and What’s Artificial?
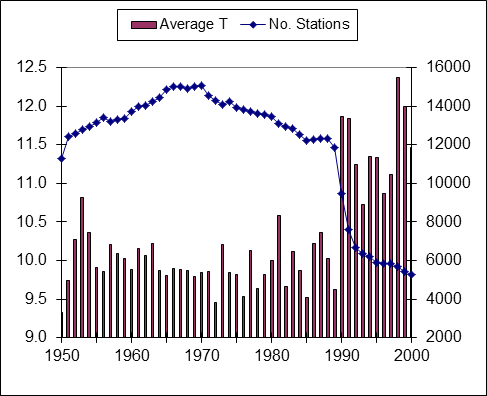
Claim 7. Temperatures have risen faster in recent years than ever before.
Truth: Remember that there is no such thing as a global temperature. It is the average of all of the reporting stations all over the world. The rapid rise in temperatures in the 1990s directly coincides with a decrease in the number of Siberian weather stations reporting due to the break-up of the Soviet Union.
Number of World Reporting Stations & Average Temperatures (vertical bars)
Source: Ross McKitrick, http://www.rossmckitrick.com/
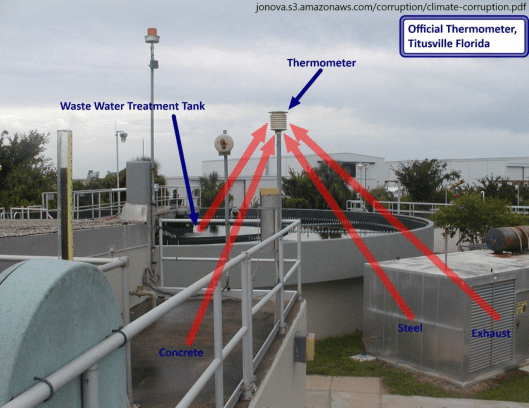
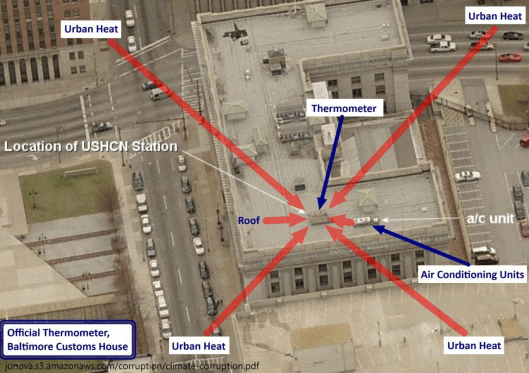
Additionally, the “hottest year on record”, 1998, was an El Nino year so it was naturally hotter than the years just before and after. Another “cause” of rising average global temperatures is the urban heat island effect. Cities are hotter than rural areas. Many of the reporting stations that were once in undeveloped rural areas have experienced either suburban or urban development, or the stations have been moved to more urban settings. It is well documented that some have been, seemingly intentionally, relocated near or at heat sources such as paved parking lots and air conditioners.
One reason for relocation near buildings or other structures could be that new automatic-reporting equipment needs to be connected by cable. Rather than dig up parking lots or roads to install units in a grassy or protected area, many have opted to locate them where they can be directly connected without involving costly excavation, although such sites do not meet the stated requirements. Instead of excluding data from stations that are poorly situated, a convoluted mathematical algorithm (scheme) is used to “correct” it to presumed pre-industrial levels. See illustrations below.
Urban area plus mathematical correction algorithm equals pristine nature
Source: “The Influence of anthropic surface processes and inhomogeneities on gridded global climate data” Slide 21, Powerpoint presentation to the American Chemical Society, Denver CO via Webinar, August 28 2011 by Ross McKitrick, Department of Economics, University of Guelph, Guelph ON Canada http://www.rossmckitrick.com/general-overviews.html
Proposed rating of reporting stations with regard to surroundings:
Figure 3. U.S. Historical Climate Network (USHCN) station exposure at sites representative of each CRN class: CRN 1, a clear flat surface with sensors located at least 100 m from artificial heating and vegetation ground cover <10 cm high; CRN 2, same as CRN 1 with surrounding vegetation <25 cm and artificial heating sources within 30 m; CRN 3, same as CRN 2, except no artificial heating sources within 10 m; CRN 4, artificial heating sources <10 m; and CRN 5, sensor located next to/above an artificial heating source.
Source: “Analysis of the impacts of station exposure on the U.S. Historical Climatology Network temperatures and temperature trends,” Souleymane Fall, Anthony Watts, John Nielsen‐Gammon, Evan Jones, Dev Niyogi, John R. Christy,5and Roger A. Pielke Sr., Journal of Geophysical Research, Vol. 116, D116, D14120 doi:10.1029/2010JD015146, 2011
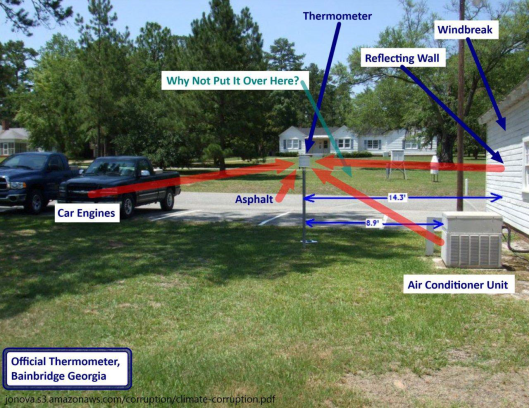
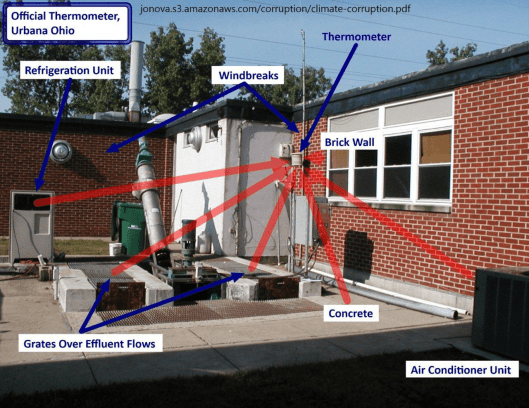
See other examples below of poor placement of temperature reporting stations from “Is the Western Climate Establishment Corrupt?” The public might not understand the science, but they do understand cheating, Dr. David Evans, 11 Nov 2010 (last updated 28 Feb 2011) Web Address: jonova.s3.amazonaws.com/corruption/climate-corruption.pdf
Claim 7 source is book, Perverted Truth Exposed: How Progressive Philosophy has Corrupted Science, 2016
The New Pause lengthens by two months to 6 years — Watts Up With That?

The miniature La Niña that has recently ended is now working its magic on lower-troposphere temperatures. The UAH monthly global mean lower-troposphere anomalies for April 2021 show a further drop, lengthening the New Pause by two months, from 5 years 10 months last month to 6 years this month.
The New Pause lengthens by two months to 6 years — Watts Up With That?
Pennsylvania Legislature Hearing on US Presidential Election Irregularities
Shocking testimony by many sworn and qualified witnesses that indicate fraud and other irregularities.
The video is almost 4 hours long. See Full Transcript Below:
Nov 26, 2020
Pennsylvania Senate Republican Lawmaker Hearing Transcript on 2020 Election

Rev › Blog › Transcripts › Congressional Testimony & Hearing Transcripts › Pennsylvania Senate Republican Lawmaker Hearing Transcript on 2020 Election
Senator Argall: (04:38)
The appointed time having come and gone, let’s ask our state senators and our state representatives to join us. Today’s Senate Majority Policy Committee hearing is the second of our hearings this year designed to help us learn more about the conduct of the 2020 elections in Pennsylvania. Today’s hearing was requested by Senator Doug Mastriano, who in addition to serving the 264,000 residents of the 33rd district in Adams, Cumberland, Franklin and York Counties, also serves as the Chair of the Senate’s Intergovernmental Operations Committee. As Dr. Mastriano can attest, this subject is an excellent example, not of the normal layer-cake model of intergovernmental operations, but of the marble cake model. We have examples of local government, county government, state government, and federal government, all very much involved in the design and conduct of an election, and as we’ve seen, not always in a logical, rational, or understandable model. I have frequently reminded the students in the college classes which I have taught, as well as more recently, our own governor, that senators and representatives and governors are not kings, we’re not judges, we’re not dictators.
The GOP Pennsylvania Senate Policy Committee held a public hearing to discuss 2020 election issues and irregularities on November 25 in Gettysburg, PA. Rudy Giuliani was in attendance at the hearing for Trump’s legal team, and Donald Trump called in on speakerphone. Read the full transcript of the hearing here.

Senator Argall: (06:21)
We are agents of public opinion elected by the people for a fixed term as lawmakers, to provide oversight on behalf of the public, over our government agencies, and of course, as lawmakers. We’re here today because we have all been receiving massive numbers of phone calls and emails and personal comments when we’re out walking our dogs in our neighborhoods, about the conduct of this election. My office has received a record number of contacts, over 25,000 phone calls and emails as of yesterday. We all know that Pennsylvania Secretary of the Commonwealth changed the guidance to election officials the day before the November 3rd election, which added a completely new element of doubt to our process, issues that should have been resolved by the legislature, as recommended by Supreme Court Justice Max Baer, were instead carried out by the Secretary without the approval of the Senate or the House.
Senator Argall: (07:22)
This committee held a similar event in January of 2020, on recent changes to our state’s election laws. At that time, the Secretary of the Commonwealth assured us that this process would be carried out smoothly. The large number of complaints which we have received from our constituents, tells us a very different story. That’s why we’re here today, again, as agents of public opinion. Senator Mastriano and his staff have assembled a number of witnesses who will share their experiences with us regarding the conduct of this year’s election. We look forward to listening to that testimony, but before we turn there, I want to introduce my good friend, our Caucus Administrator, and as of December 1st, our incoming Majority Leader, congratulations to Kim Ward. Thank you for joining us. Please Kim, your statement.
Senator Kim Ward: (08:15)
Thank you, Senator Argall, and thank you, Senator Mastriano, for inviting me to participate in what I think is very, very important to the people of Pennsylvania. As Senator Argall has said, we have been inundated with calls, and emails, and messages in our social media. People are not feeling good nor confident about the process and the results because of that process. The President and his team deserve… This is the President of the United States, not the President of your middle school, school board. We need to make sure that they have the room that they need to explore every avenue, so when this is over, we know that the process, what worked in it, what didn’t work in it, and what we need to do to fix it, and what we may need to do to address it. This has been very, very exhausting and hard on so many in Pennsylvania. And I look forward to listening to what the testifiers and Mr. Giuliani have to say to us today. Thank you very much again, and thank you. Thank you, Senator Mastriano.
Senator Argall: (09:24)
Thank you, Kim. Senator Mastriano, please.
Senator Mastriano: (09:26)
Thank you, Senator Argall and Senator Ward. Thank you for being here and supporting this here. And welcome everybody to this most historic occasion and this most historic town and what happened here in 1863. I think today we’re going to see a turning of the tide because we have not really heard the truth of the arguments made on the other side and what happened and what we’re dealing with, a government and leadership in Harrisburg that wants to close their ears to what’s happened during this election, and sadly, many in the media that are complicit and want to write off what happened. For me, on this battlefield and remembering what happened 137 years ago, and especially 157 years ago last week, where Abraham Lincoln gave his most famous address.
Senator Mastriano: (10:09)
And I think his final sentence captures why we’re here today. He said, “That this nation, under God, shall have a new birth of freedom, and that government of the people, by the people, for the people, shall not perish from the earth.” And everything is at stake in what happened during this election cycle, everything, the Republic is at stake. This is no game for us. And for any veteran in this room here, who wore the uniform, such as Brigadier General Scott Perry, thank you for being here sir, put their lives on the line here fighting for our country, and to see that there is a group in this state and country willing to throw away our valuable and precious freedoms here for power. You know, for me… There we go. And Mr. Mayor, it’s going to be a fantastic hearing today. Despite pleas from our citizens, the governor refuses to even consider that there was any shenanigans in the elections here in our state, a state of 13 million people. And he wants a discount because his guy won, that nothing bad happened, and that’s just unacceptable.
Senator Mastriano: (11:20)
If there’s any hint of fraud out there, we need to investigate. Any governor serving the people of this commonwealth, would put aside his petty politics, would put aside his petty politics and find out the truth, if anyone was defrauded. And at this point here, he’s unwilling to do that. Let me point out some hard facts here. We are in Adams County named after our second President, John Adams, who famously said, “Facts are stubborn things.” Let me lay out several of these inconvenient truths as Al Gore would have us think. There have been many allegations of voting law violations across the state and a governor serving the people would have to move heaven and earth to ensure no one was defrauded, but yet he’s not moved to action. And even his Secretary of State of course, says that there was no shenanigans of great concern.
Senator Mastriano: (12:04)
And I will remind everyone that I was a no-vote on the confirmation of Kathleen Boockvar four times because I interacted with her. I asked her, “Why are elections in Afghanistan more secure than in Pennsylvania?” And she sat there and blinked and couldn’t give me a straight answer that elections are more safe and secure in a war-torn country, devastated by conflict since 1979. What is going on here? The place where this all started in 1776 and we can’t get an election right? You got to be kidding me, here. As a result of her inaction, the governor’s inaction, refusal to even look into any of the allegations and to discount the very essential freedoms of our citizens, we are here today to try to find out what the heck happened in the election.
Senator Mastriano: (12:53)
And likewise, our Attorney General, our senior law enforcement official here, instead of being focused on making sure things are lined up and kosher, before one vote was counted in Pennsylvania, the day before the election, declared Biden the winner. There’s nothing to see here. Could you imagine if the shoe was on the other foot? Would the media be so gracious and merciful and kind if it was a Republican? Absolutely not. Houston, we have a problem here. One of the most troubling things in this whole endeavor here, and it’s not just because of the COVID, it’s the lack of transparency and accountability. So we’re here to start shedding light on the darkness. And then of course, we have a Supreme Court that rewrote election law, Act 77 has been painted as a villain. Okay, we could debate that. But the real problem was, is when the Pennsylvania Supreme Court decided they’re going to write legislation and rewrite our law. And because of that, obviously Pennsylvania, we’ve got a lot of problems there and that opened the door to all the shenanigans and abuses and folly that we’re dealing with here in this state this day.
Senator Mastriano: (14:04)
What’s going on here? Thousands of people from across the Commonwealth have reached out to us, tens of thousands, asking and demanding action, they deserve it. And as a result of the inability of our Executive Branch to do their job, we’re stepping in here. We’re co-equal members and we’re going to do our job. We’re looking for transparency and truth. There’s going to be no grand-standing here, we’re after facts. And we’re going to have a good layout here of what happened. And you’re going to have to decide, good people of Pennsylvania, on what happened and whether there’s a strong case we made or not. The forgotten men and women of our great state feel betrayed by their government and I’m with them, I feel the betrayal as well. We undertake these proceedings today to find out what happened and then hopefully to come up with an approach where that never happens again. And the issues are galore, you’re going to hear about poll watchers being denied access, where election software vendors refuse to testify before the General Assembly, what do they got to hide?
Senator Mastriano: (14:59)
Do we expect the people to trust their government? Then we’ve got to earn their trust. And this is no game. And the very Republic very much is at stake. And anyone who loves this country has to put aside their petty, partisan politics and allow the light to shine where it is. And we’re going to find the truth and celebrate it. As a soldier, and now as elected member of the State Senate, I’m not going to stand aside and neither of the members around me are as well. We’re going to fight the good fight for freedom and secure our Republic. Too many good men and women have gone before us, who have given their lives here, and to cast it aside now for power play, is unacceptable, it’s not going to happen. Thank you. [inaudible 00:15:39]. We do our great men and women in uniform, and those who lay down their lives and gave the last full measure of devotion, a great disservice if we stand aside and allow bureaucrats and corrupt politicians to steal their voice and maybe even steal an election, we’ll find out.
Senator Mastriano: (15:58)
John 8:3, thank you, John 8:36 says, that if Jesus sets you free, you’re free indeed. We’re going to walk as free people in Pennsylvania, this is where it all started. We choose this day to walk as free men and women, and honor the sacrifice, not only of Jesus on Calvary, but also in honor of the sacrifice of brave men and women in uniform who fought for and secured our freedoms. We will be relentless in our pursuit of the transparency of accountability and truth. The time for dithering, politics, and games is over. The time for truth and justice is now. In conclusion, as Benjamin Franklin is leaving Constitutional Hall in 1787, he was approached by Mrs. Powell. And Mrs. Powell ran up to him, we surmised she knew him personally, and she said, “Well, Mr. Franklin, what do we have, a monarchy or a Republic?” And he said, “A Republic, if you can keep it.” This is our time to keep this Republic. Thank you and God bless you.
Senator Argall: (16:54)
Thank you. Thanks, Senator Mastriano. [crosstalk 00:17:12]. We turn now to the introductions of our House and Senate members. Let’s begin with our representatives here at this end of the table. Gentlemen, ladies.
Representative Dave Zimmerman: (17:38)
Is it on? Yeah, Representative Dave Zimmerman, serving the Northeast part of Lancaster County.
Representative Greg Rothman: (17:47)
Representative Greg Rothman, 87th District, Cumberland County.
Representative Mike Jones: (17:55)
Representative Mike Jones, 93rd District, York County.
Representative Paul Schimmel: (18:01)
Representative Paul Schimmel, portions of Franklin County.
Representative Rob Coffman: (18:06)
Representative Rob Coffman, 89th District, Northern Franklin County.
Representative Stephanie Borowicz: (18:13)
Representative Stephanie Borowicz from the 76 District. I’m honored to be here fighting for freedom, so thanks.
Senator Judy Ward: (18:24)
Hi, Senator Judy Ward from Blair County, representing Blair, parts of Huntington, parts of Huntington, parts of Franklin County, part of Cumberland County, and all of Fulton County.
Senator Argall: (18:40)
You’ve already met Senator Kim Ward. I believe you know Senator Mastriano.
Senator Mario Scavello: (18:51)
Good afternoon, I’m Senator Mario Scavello from Monroe/North Hampton County, and I could see New Jersey from my district.
Senator Mike Regan: (19:00)
Senator Mike Regan from Cumberland and York County.
State Representative Dan Moul: (19:07)
Good afternoon, State Representative Dan Moul, welcome to my 91st Legislative District.
Senator Frank Ryan: (19:17)
Representative Frank Ryan from Lebanon County, [breakaway 00:00:19:21]. Thank you.
Senator Argall: (19:25)
We are also joined on Zoom, Senator Brooks and Senator Hutchinson from Northwestern Pennsylvania, Senator Yaw from the Williamsport area, Senator Stefano from Southwestern Pennsylvania, Senator Laughlin from Erie, Senator Martin from Lancaster County, Senator Pittman from Indiana County, Senator Pat Brown from the Lehigh Valley. Also, my job as Chairman is to remind our panelists to keep their microphones muted until it is your turn to speak, and also remind everyone to strictly observe our time limits. One other reminder to our senators and witnesses at the request of our Senate attorneys, this is a legislative hearing. Our purpose is to listen to the complaints of our constituents, the oversight of government agencies, and the need for possible legislation. We all need to conduct ourselves accordingly. With that being said, Senator Mastriano, I believe you want to introduce our first witness for 15 minutes and we will hold, to the representatives and senators, so you understand, we will hold all of our questions until the conclusion of the sixth panel.
Senator Mastriano: (20:37)
I would just simply say, welcome to America’s Mayor. Thank you, Mayor Giuliani, for being here. [crosstalk 00:20:53].
Rudy Giuliani: (21:00)
Senators, representatives, Senator Mastriano, and all the distinguished members of the legislature of Pennsylvania, we are very, very honored and very appreciative that you are giving us an opportunity to be heard, which we’ve been denied almost uniformly by the media and by legislatures elsewhere. All we ask is that you listen to the facts that we’re presenting and then evaluate it. I can’t help but note that we’re doing it here in Gettysburg. Over 156 years ago, the fate of our country hung in the balance, right here on this hallowed ground for three relentless days of bloody, close combat between two armies that lost over 50,000 casualties. The fate of our Republic was really decided that we’d be one nation, one union, one government sharing values that are enormously important to us. And I don’t want to overstate it, but I do believe that those values are at stake, not only in this election, but in the way this election was conducted, and what we’re going to do about it. Because if we allow elections in the future to be conducted the way this election was conducted, we will have lost our democracy, our representative democracy.
Rudy Giuliani: (22:27)
During the course of this election, we’ve come pretty close to losing our right to free speech. There’s been censorship that I’ve never seen before, of an incredible nature by big tech, big networks, big companies. They only allow one side to be heard and they refuse to allow the other side to be heard. It’s almost as if they’re afraid the American people, if they should learn these facts, will find out just who they are and what they’re about. This voter fraud that took place, which as you will see from the witnesses that we call, had several dimensions to it, several different ways in which it was done. The most dangerous thing is, it is very, very similar in at least six states that we’ve been able to study. In other words, what we’re going to describe to you with these witnesses, happened in roughly the same way in Michigan, Wisconsin, Nevada, Arizona, and Georgia. Primary device, were the mail-in ballots. You know there was a fierce debate over whether we should have mail-in ballots in the first place.
Rudy Giuliani: (23:52)
Many scholars, many experts, always felt that mail-in ballots were very dangerous because they’re very easy to forge, it leads to more defrauding. We will warned by Justice Souter, among others. We will warned by President Jimmy Carter and former Secretary of State James Baker, in a report that they did on how to make elections more secure. They warned us that the one thing to do is do not go to general mail-in voting because every place it’s been used, it’s led to tremendous fraud. And that was reiterated, believe it or not in an article in the New York times in 2012, an article they have now forgotten they wrote, that explained the same thing. And I think what you’re going to find as you study your mail-in ballot procedure here in the Commonwealth of Pennsylvania, and then in the six other places, this was a terrible mistake and it allowed the leadership of a party that had become pretty expert at voter fraud, to really go wild.
Rudy Giuliani: (25:06)
Witnesses we present are going to first show you that, in the case of Philadelphia, and in the case of Allegheny County, and one or two other counties, the mail-in ballots that were received, were not inspected at all by any Republican, they were hidden from Republicans. In the case of Philadelphia and Allegheny County, I can’t be absolutely certain, but I do believe the witnesses will show that a Republican never got to see a single ballot. Now, you know how important that is to determining whether it is a valid ballot or not. There’s only one time you get to do that and that’s when you separate the envelope that possesses the verifying information from the ballot. The moment you separate them, you can no longer verify. You can’t go back and recount, you can’t go back and check paper ballot against the machine ballot, because it’s a private ballot, it’s an anonymous ballot, we want to keep it that way.
Rudy Giuliani: (26:11)
The only thing that identifies it, is that outer envelope. At that moment, when they’re separated, gone forever. That’s the moment at which inspections have been allowed, time immemorial, in America, Pennsylvania, all of our other 49 states. Several of the witnesses here have been doing this for 20 or 30 years, they’ve never heard of a situation in which the mail-in ballot was just put in without allowing a Republican, a Democrat, even third-party members to take one look at it and object to it, for the very reason that it’s too prone to fraud. Think about this, in your state, Republicans were uniformly not allowed, kept out, put in chutes like they were cows, to keep them away from seeing these ballots. Never happened before. Not only that, the same thing was done in Michigan, the same thing was done in Wisconsin and on and on and on.
Rudy Giuliani: (27:12)
What’s the chance that, on the morning of November 3rd or 4th, when they started the count, that in each one of those places, the Democrat leadership of these highly-controlled Democrat cities, that have some history for corruption, and in the case of Philadelphia, a long history of voter fraud, I could show you the convictions, I don’t think I have to. What are the odds that they’re all going to wake up with the same idea? After years and years of always examining together, absentee ballots, all of a sudden, in a year in which we have a couple million of them per state, we’re not going to allow any Republicans to see them. The person in Philly figures that out, Pittsburgh, Detroit, Milwaukee, Las Vegas, Nevada, or is it more likely that this was a common plan that maybe started with the whole idea of having mail ballots, because it gives you a much wider range to cheat?
Rudy Giuliani: (28:14)
When you had just a small number of absentee ballots, like 400,000, you have a certain range that you can cheat. When you have 250 or 2.5 million, you have a much bigger range to do that. When you hear that testimony about not being allowed to see the ballot, have to understand it’s much more important than just that individual, just that individual ballot. On election night, when I went to sleep, maybe when you did, President Trump was leading in your state by somewhere around 700 to 800,000 votes, depending on when you went to sleep. That’s a huge number of votes, 65% of the vote had been cast. Under normal circumstances, like if this were a fair media, your state would have been called for Trump. Virginia was called with 10% of the vote, it turned out to be separated by 1%.
Rudy Giuliani: (29:09)
I think we may have actually won Virginia, but that’s another battle. Michigan, we were ahead by 300,000 votes, Wisconsin, more. Georgia, we were down to 90% and ahead. What are the odds that they all switched overnight? They just switched by the next day. I think you’re going to see how that happened. And I think there are a couple of statistics that you have to really closely look at. I’ll just mention them and then we’ll move on to the witnesses. We have calculated, and the evidence will show, that there were 682,770 mail-in ballots that were entered into your votes, in just Allegheny County and in Philadelphia, that were not observed by any single Republican. Those ballots could have all been for Joe Biden, they could have all been for someone else, they could have had no identifying data, they could have been from the same person. There could have been multiples of them, there could have been no name on them.
Rudy Giuliani: (30:17)
We have no idea if that’s true and it will be very hard now to kind of put them together. We could ask and you could subpoena all of the outer envelopes. It’d be very interesting to take a look at the 682,770 outer envelopes. It would be very interesting if they were kept, and it would be very interesting to see how many of them weren’t filled out. But in any event, under the law of your state, which is set by you, those ballots are illegal. The judge mistakenly in his opinion, said that we want to disenfranchise 6 million people. We don’t want to disenfranchise anyone, we want to disqualify 682,000 votes so that 74 million people are not disenfranchised, because that’s what happened by the cheating that went on here.
Rudy Giuliani: (31:10)
I’ll give you one other enormously puzzling statistic. You sent out in the State of the Commonwealth of Pennsylvania, 1,823, 148 absentee or mail-in ballots. You received back 1.4 million, approximately. However, in the count for President, you counted 2.5 million. I don’t know what accounts for that 700,000 difference between the number of ballots you sent out and the number of ballots that ended up in the count. That number, 2,589,242 was on your government website until yesterday. And yesterday, it was removed without explanation. I’m going to be very interested in hearing what the explanation is. And I can’t imagine you could possibly certify without knowing the…
Rudy Giuliani: (32:03)
… Is, and I can’t imagine you could possibly certify without knowing the explanation to that, as well as the explanation to the 22,686 mail-in ballots that were returned on the day they were mailed. That’s a trick. How about 32,591 were returned the day after they were mailed? Another 20,000 were returned before they were mailed. Of course, I think this is a kind of a low count, and I guess, the crooks in Philadelphia are disappointed in this. They only submitted 8,021 ballots from dead people, mail-in ballots for dead people. Probably easier for dead people to submit mail-in ballots than it is to vote in person. You had about 30,000 of those. We’re checking the records of the cemeteries around Philadelphia.
Speaker 1: (33:01)
[inaudible 00:33:02].
Rudy Giuliani: (33:04)
You have 4,984 mail-in ballots that were never requested, and on, and on, and on, and on. Your election, because of these two counties and maybe one other, is a sham. It’s a disgrace to your state. And finally, I don’t need to remind you of this. I think I need to remind America of this. The election for president of United States is not run by the governor of your state. It’s not run by your election commissioner. The United States Constitution makes it clear who has the responsibility for running this election? Article ll, Section 1, Clause 2 of our Constitution doesn’t say that ABC gets to call the winner or CNN. It says, ” Each state shall appoint in such manner as the legislature thereof may direct, a number of electors.” It’s the state legislature that controls this process. It’s your power. It’s your responsibility.
Rudy Giuliani: (34:23)
And I think you know, and you have to convince the rest of your members, Republican and Democrat, they owe that to the people of your state, and they owe that to the people of the United States. Because if this happens without consequence, if they can just enter 600,000 some odd mail-in ballots without allowing a single Republican to view it, what’s to say that next time, they won’t do a million or 2 million? I know crooks really well. You give them an inch, and they take a mile. And you give them a mile, and they take your whole country. So, now, we’ll proceed with the witnesses.
Senator Argall: (35:14)
Thank you. Thank you very much, Mr. Giuliani. I’ve been informed that we have also been joined on Zoom by Senator Kristin Phillips-Hill, Representative Dawn Keefer, Representative Barb Gleim from central Pennsylvania, Representative Daryl Metcalfe from Butler County, and Senator Langerholc from Johnstown area. Senator Mastriano, would you like to introduce our next panel for the, it looks to me like several people for 15 minutes as well.
Senator Mastriano: (35:46)
Yes. Thank you, Senator Argall. Our witnesses, first off, Jen Ellis, you have any comments for the floor?
Jen Ellis: (35:55)
Thank you.
Senator Mastriano: (35:58)
Thank you. Legal advisor to President Trump.
Jen Ellis: (36:00)
Thank you very much. And I’ll reserve my comments for the end, and I would just echo everything that the mayor so eloquently stated. Thank you very much.
Senator Mastriano: (36:09)
Thank you. This is the revised list here. We’d like to call forth Justin Kweder from Philadelphia. Justin, are you here?
Justin Kweder: (36:18)
I’m here.
Senator Mastriano: (36:19)
Okay, outstanding. If you can come up to the table, please, my friend. And Kim [Peterson 00:36:24] out of Pittsburgh. Kim, are you here?
Kim Peterson: (36:27)
Yes. Right here.
Senator Mastriano: (36:27)
Come forth, please. We have a seat for you here. I guess [inaudible 00:36:32].
Senator Argall: (36:31)
We were going to do four.
Senator Mastriano: (36:35)
Okay. [Leah 00:04:36]? Leah [Hoops 00:04:38]. Where are you, Leah? Please come forth. Kayla, could you push those two chairs over? Thank you. And that American flag is mine. If you set it on the table, I’d appreciate it. Thanks. And then Greg [Stenstrom 00:36:54] from Delaware County. You’re zooming in, I believe.
Speaker 2: (36:57)
No, he’s here.
Senator Mastriano: (36:57)
Oh, he’s there? Yeah, of course. I met you. Come on over, brother. [inaudible 00:37:03] Okay.
Senator Argall: (37:02)
Okay. If you can among yourselves, please divide up the next 15 minutes. And again, we’ll hold off questions until the end. Please begin.
Speaker 3: (37:22)
[inaudible 00:00:37:20]. Okay, he’s next to her. Okay.
Justin Kweder: (37:24)
I’ll start. Thank you, Senators and Representatives for inviting me to this important hearing. My name is Justin C. Kweder, K-W-E-D-E-R. I am a resident and registered voter in the city and county of Philadelphia. I’m an attorney. I am barred here in the Commonwealth. I am a volunteer certified GOP canvas observer. I was present at the Philadelphia Convention Center during the pre-canvas, canvas processing, and counting of the ballots cast in this year’s general election. I was first present as an observer at the convention center and at the count on election day. I returned as a volunteer observer to watch the process every day for the next 10 days. I’d approximate I was there for about 85 hours over those 10 days.
Justin Kweder: (38:14)
Part of the reason that I kept going back was so that I could authoritatively speak about what I saw. And again, thank you for letting me be here because what I saw was problematic, to say the least. I can tell you that I am an eyewitness to many issues and irregularities that were observed in Philadelphia with the processing and counting of the vote. I was a witness to too many issues and irregularities to cover in a brief opening statement. And as such, I will now focus on just two issues that I did personally observe.
Justin Kweder: (38:47)
First, the Philadelphia Board of Elections processed hundreds of thousands of mail-in ballots with zero civilian oversight or observation. The mail-in ballots were handled processed, opened, and counted in a Hall F of the convention center. Hall F is a vast room, approximately 350 feet by 350 feet. That’s about 120,000 square feet. The Board of Elections erected a fence approximately 50 feet into the hall that ran the length of the room. All observers were corralled behind the fence. More than a hundred board workers processed and opened mail-in ballots on the other side of the fence. These masked workers were arranged throughout the 120,000 square feet at a distance from the observers of about 10 feet to more than 200 feet away from us. Unlike what some may have reported in the media, the vast majority, and I’d estimate it at, at least, 96% of the board workers were processing mail-in ballots 15 to 200 plus feet from us.
Justin Kweder: (39:54)
Due to the distance of the workers from the erected fences, it was impossible for me or any observer to see what the workers were doing with any type of specificity. The observers were not able to challenge any decision or determination being made about the processing of these mail-in ballots. No civilian oversight over hundreds of thousands of mail-in ballots, zero opportunity for observers to observe, challenge, or inspect the mail-ins being opened. I find this to be an issue and to be irregular.
Justin Kweder: (40:26)
Move on to the second point, the pink highlighted duplicate ballots, which I haven’t heard about, at least not in the media. As the weeks progressed, the observers were informed that the board was going to be duplicating damaged mail-in ballots that could not be read by the scanners. I was told that there were more than 5,000 of these damaged ballots. I was also told that the actual number was unknown. It could be much higher. The process for duplicating these ballots was for two to work as a pair, one worker reading out loud what was marked on the original damaged ballot, and the second worker using a pink highlighter to mark the choice on the duplicate ballot.
Justin Kweder: (41:06)
The board’s plan, then, was to run the pink highlighted ballots through the scanners where they would be counted as votes. The board workers did this pink highlighter duplication work over the course of a couple days until thousands of ballots were duplicated with these pink highlighters. On Thursday, November 12th, the observers were informed that the pink highlighter could not be read by the scanners. They all had to be done again. All of the duplicated mail-in ballots had to be fixed. And the Philadelphia Board of Elections’ solution to this problem was to give the workers who were working alone, individually, stacks of hundreds of what amounted to blank mail-in ballots. And the workers, individually, were to fill in the correct highlighted ovals with dark pen, no observation.
Justin Kweder: (41:54)
They were marking thousands of blank mail-in ballots. The workers did this double recreation work for hours before the observers realized what was going on because we weren’t told what was going on. Only when the observers confronted the deputy commissioner about this irregularity was a system set up where the workers would show the observers each double recreated mail-in ballot by flashing it at them for a second, from a distance of 6 to 20 feet away, for as good as that was. Thousands of mail-in ballots were ultimately counted in this way. Again, I find this to be an issue and to be irregular. As Senator Mastriano recently said, elections are a fundamental principle of our democracy. Unfortunately, Pennsylvanians have lost faith in the electoral system and quote… And let me add a quote that Philadelphia City Commissioner of Elections Al Schmidt just made when he said, about these recent elections in Philadelphia that, “These were the most secure and transparent elections in Philadelphia’s history.” Now, I wasn’t around for every election in Philadelphia’s history, but I can tell you, as an eyewitness, for 10 days, for more than 80 hours, what I saw was not a secure and transparent election. There are major concerns about the legitimacy of hundreds of thousands of ballots that were counted in Philadelphia.
Justin Kweder: (43:25)
Excuse me. All right. Let me finish with this. May I suggest to you that any restoration of faith on the part of Pennsylvanians in our electoral system can and will only be accomplished after an investigation into this election, only after the law is properly applied to this election. The idea that we can just move on by making sure that the law is properly applied in the next election is, in my opinion, unacceptable. The issues, irregularities, and illegitimacy of what I saw leads me to conclude that the laws of the Commonwealth and of the United States were not being followed in Philadelphia when hundreds of thousands of ballots were counted in this year’s general election. Action must be taken now to maintain our free and fair elections and to preserve and protect our election’s integrity. I look forward to answering any questions that you may have. Thank you.
Kim Peterson: (44:30)
Good afternoon. My name is Kim Peterson, and I appreciate that I’m here today. Thank you all for having me here today. I’m from Pittsburgh, Pennsylvania. And I am here to tell you what I witnessed as a canvasser, where the warehouse where they opened the mail-in ballots and separate them from the envelopes as we have been discussing. The afternoon of Election Day, I went down to the warehouse to be a canvasser, as a Republican representative, to be able to watch them open the mail-in ballots. And as the gentlemen, Justin, just described, as I was led back to the area where we were able to view them opening the mail-in ballots, we were kept in a corral that was at least 15 to 20 feet from any of the representatives. And that was the closest people opening the ballots. There were people that were 50, 100 feet from us. The closest was 20 feet, about, and you could not see, at all, the envelope, the ballot itself, where they were stacking them, anything that was required that we were able to be able to see.
Kim Peterson: (45:43)
There were also monitors up around the area. The monitors were pathetic, to be honest. They were fuzzy, looked like they were using old technology, as far as… We have the most clearest televisions created in the world now. And these were probably from the ’60s. You’re looking at all these multiple cameras, screenshots put together of people opening ballots, but you could not even see at all or witness anything if any misdoing or anything going wrong. It was very discouraging, on my part. I was very much looking forward to being a part of that, that day, and representing myself and Pennsylvanians in a fair election. And I was, I guess, discouraged about it. I love this country, and I love Pennsylvania. I was not born and raised in Pennsylvania, but I have been here for 30 years, and it is very important to me that this election be fair. And thank you again for having me.
Leah Hoops: (47:05)
Excuse me. My name is Leah Hoops. I’m from Delaware County, Pennsylvania. I was a poll watcher on the day of election, and I was also present at the counting center in Chester, Pennsylvania. First and foremost, thank you to this committee and all those involved, and especially, Senator Mastriano and former mayor, Rudy Giuliani. To our fearless and brave president, thank you for being our shield and putting us first, and I am forever grateful. It is an absolute honor to be a part of this hearing and to finally have an opportunity to speak about what took place in Delaware County, Pennsylvania. I feel, as an American, that is my duty to help protect the integrity of our elections, not just for me, but for every American. There are many who have lost life and limb for my right to vote. I want to give a brief introduction and history of how I got involved in this election.
Leah Hoops: (47:51)
Four years ago, Donald Trump came down an escalator, and I knew that he was here to take back our country. I was born and raised in Delaware County in a conservative home. I was taught from an early age to love thy neighbor, defend your country, and by all means, always speak the truth. I took those values throughout my life and applied them in every avenue possible, which brings me up to the present. And about 18 months ago, I became a volunteer and assisted with a campaign for a magisterial judge. In that process, I was approached by my current chairman and became an appointed committee woman for the Bethel township Republican party. In conjunction with my committee position, I also started volunteering for the Trump campaign, registering voters, and assisting with events. During this time, I started a watchdog group and reached out to the Thomas More Society.
Leah Hoops: (48:36)
Now, associates, we’ve been focusing our efforts in election integrity. Our greatest efforts have been in following closely, the newly-elected, and completely Democrat-run Delaware County Council, and the Democrat Majority Election Board. This also includes any and all contractors, support staff, and any key players involved. What we found was actually concerning. Not only was private grant money used from the center for tech and life owned by Google and Mark Zuckerberg, but pop-up voter sites were also approved. These pop-up voter sites were placed in heavily Democrat cities, including Chester and Upper Darby, Pennsylvania, in which case the grant money from the CTCL was used to pay for electioneering. It was literally a one-stop shop. Walk in, apply, get your ballot, submit, and you were out the door. But where this didn’t take place was in heavily Republican and independent areas. Let’s also make note that the voting machine warehouse supervisor is a Bernie Sanders delegate, who was also solely responsible for every scanner, machine, V card, and all machines with absolutely zero experience in this area. The real story is the $2.1 million spent to move a counting center [Inaudible 00:50:18] Okay. Hello? Okay. Sorry. Sorry. I forgot where I was. Okay, so the real story is the $2.1 million spent to move the counting center from the courthouse to Media, Pennsylvania, which is something that has been done for decades, to the wharf in Chester, Pennsylvania. Let me explain to you the layout of the counting center. It is in the middle of a huge parking lot, which sits back on Seaport Drive next to loading docks. It has multimillion dollar connections to the company, Power HRG and Subaru Park Soccer Stadium. The counting center was on the first floor, in which there were multiple places to bring in ballots in and out of elevators in many rooms in which workers had access. I was there for three straight days.
Leah Hoops: (50:56)
What became of concern was the backroom, which had no observers, no line-of-sight or transparency into the process. There was no cooperation, complete resistance from election night and every day after. It took until our lawyer got an injunction to get into that back room in which pre-canvassing was transpiring. Even with an injunction, which was a joke, we were granted five minutes every two hours. And the setup was sitting in a chair 20 feet from any physical ballot. I truly wish I had enough time today to recreate what I witnessed and felt during those moments. But we did not gather here today just for me and my experience, but rather a collection of experiences from Americans like myself. What I hope to achieve is for the public to understand is that we have stuck our necks out, have been intimidated, threatened, bullied, have spent countless hours away from our families, friends, and jobs.
Leah Hoops: (51:51)
We have signed affidavits under penalty of perjury, which should be consideration enough to know that this is a very serious issue. I’m here for one thing only, and that is to speak the truth. This is not about party. This is about my country. Every American deserves transparency, truth, and be able to question those in power without fear of intimidation, bullying, or backlash. I hope this committee takes action, if needed, and justice will be swift to anyone involved in fraudulent activity. The Republic is angry, disgruntled, tired, beaten up, and ready to defend this country.
Audience: (52:27)
Yes.
Leah Hoops: (52:27)
Thank you. Without election integrity, we are just another Banana Republic. I thank you again for your time, efforts, and service. And it is an honor to be here and to be part of a historical moment. Thank you.
Gregory Stenstrom: (52:54)
It’s a tough group to follow. My name is Gregory Stenstrom. I’m from Delaware County. I’m a father, a family man. I was a former commanding officer, an executive officer in the Navy. I’m a veteran of foreign wars. I’m a CEO of my own private company. I’m a data scientist. I’m a forensic computer scientist. I’m an expert in security and fraud. Leah had recruited me for this election, and I was glad. I thank you for that.
Leah Hoops: (53:24)
Thank you.
Gregory Stenstrom: (53:25)
So, for the first part of the day, I was a poll watcher in the city of Chester. And I was with just another former US Marine officer. And the two of us were the only GOP poll watchers in the city of Chester, which is about 40,000 people. Because of the consolidation for COVID, there were seven polls that we were able to make it to in 22 precincts. What we saw out there was pretty orderly and exciting. People were excited to vote. One of the things we saw out in the field was that quite a few people had done mail-in ballots. So, they came in, and they hadn’t been updated in the Delaware County database yet. So, they would come in and say, “The database is not showing me on the database. I’d like to vote.”
Gregory Stenstrom: (54:16)
One of the processes was to give them a provisional ballot, and then, they would vote provisionally. And then, later on, their vote would be sorted out. That didn’t happen. I observed, and the gentleman that was with me, observed seven different polls where the people were given a regular ballot. So, they cast a vote and put it in. We let it happen a few times. We didn’t jump all over them. It happened a couple of times. We went up. In all cases, the election judges were very forthcoming, very polite. They apologized, and they said they couldn’t do it. But that somewhat spurred me to go down to the counting center, which Leah described. It’s on the Seaport Ave. It’s a remote building, not much around it. And I wanted to take a look. All day long, I had been told that there were 10 to 20 GOP poll watchers down there, and that everything was well in hand. And out of curiosity, I decided to go down.
Gregory Stenstrom: (55:17)
I arrived at 6:00 with four other gentlemen, again, foreign military and some good citizens from Delaware County. And we weren’t allowed to get in until 11: 00 at night, and we had to get some legal help to get us in. So, it took us five hours to get upstairs. After that, what I saw is, I really think the crux of this in Delaware County is… As an expert in this, I think it’s impossible to verify the validity of about 100,000 to 120,000 votes. Now, Delaware County has got 425,000 registered voters. Approximately 300,000 of them voted. I don’t know what the exact number ended up. Mayor Giuliani nailed that number. But of that number, over 100,000 are in question in my mind. What I saw, as a forensics expert, was an election process that was forensically destructive in the manner it was conducted with the envelopes being separated from the ballots and going to the other side of the room. And the problem with that from being forensically destructive is that when you go to do a recount, okay, the machines did a pretty good job of recounting.
Gregory Stenstrom: (56:37)
So, if I have 120,000 ballots, mail-in ballots at one side of the room and envelopes at the other side of the room is still going to come out 100, 2,000 votes for Vie President Biden and 18,000 votes for President Trump. I don’t care how many times you recount those votes. The ballot’s going to come out the same every time. So, the notion of a recount in a forensically destructive process is, it doesn’t work very well. What we saw there, what I saw there was a chain of custody. In all cases, it was broken. It was broken for the mail-in ballots, the drop box ballots, the election day USBV card flash drives. In all cases, the chain of custody and the procedures that were defined by the Delaware County Board of Elections and election process review were all… They didn’t follow one.
Gregory Stenstrom: (57:28)
I couldn’t even redline this multi-page document because the entire document would be they didn’t follow any of the procedures. So, I personally observed USBV cards being uploaded to the voting machines by the voting machine warehouse supervisor on multiple occasions. I saw this personally. I brought it to the attention of the deputy sheriff, who was there, stationed, who was a senior law enforcement officer. And I brought it to the attention of the clerk of elections. I brought it to their attention. I objected. And I said, “This person is not being observed. He’s not part of the process that I can see.” And he’s walking in with baggies, which we have pictures of, and it was submitted in our affidavits. And he was sticking these USBs into the machines. So, I personally witnessed, that happened 24 times, over 24 times. We have multiple other witnesses that saw it, including Democrat poll watchers.
Gregory Stenstrom: (58:31)
And I was told the next day by the solicitors… Well, actually, not the solicitor, but the attorney that we had secured, that they said, “Every election, they leave a couple of USBs in the voting machines, and they’re brought back. And generally, the warehouse manager comes over and puts them in.” So, in talking to the US Attorney General McSwain and other law enforcement officers, I found out that was not the case, that generally, more than two is unusual. So, they denied they did it. But as of today, 47 USBV cards are missing, and they’re nowhere to be found. So, I was told personally that these 24 to 30 cards that were uploaded weren’t there. Those cards, I demanded that they… They didn’t update the vote lifetime. They only updated it about once every two or three hours.
Gregory Stenstrom: (59:33)
I demanded they updated the vote so I could see what the result was, and it was 50,000 votes. And I think as a computer scientist, an American, and a Patriot, it doesn’t matter who those 50,000 votes were. I’ll tell you they were for Vice President Biden. But what was shocking to me, as an American, as someone who has gone to sea, gone to war that that could even happen. So, several other things that came up was on Thursday, it took us three days for them to obey the court order that I secured with Leah’s help and the help of the Thomas More Society, who we thank. Incredibly good patriots. They got us in there. They got the order for us to get in and look at the back offices, which were locked, for five minutes every two hours. I went in. I was the first one allowed to go in at 1:30 on Thursday, and then, again at 3:30 for five minutes.
Gregory Stenstrom: (01:00:42)
The County solicitor had a stopwatch. On the first time, I was not allowed to touch anything. The second time, I did. What I observed in the locked room in the back office was 70,000 unopened mail-in ballots. They were in boxes of 500 stacked in neatly. The gentlemen that came in with me was a Democrat poll watcher, is a forensic pathologist, a very detailed, very dedicated man. And he took meticulous notes as well. And I verified with him, “Are you seeing what I’m seeing?” We both agreed, as GOP poll watchers and a Democrat poll watcher, that we had a witnessed 60 to 70,000. We had a little bit of a disagreement there. The problem with that was by that time, the mail-in ballots had already been counted.
Gregory Stenstrom: (01:01:37)
So, 120,000 mail-in ballots had already been counted, posted, and done. My question is, where did the 70,000 ballots go? And nobody knows. We have a picture in here of a large number of boxes that I took that were filled with what appeared to be ballots sitting by the BlueCrest machine. They were there for about three hours, and then, they disappeared. I thought it notable when I watched it, the first when taking the ballots up and down. I said, “I am an expert in fraud.” I saw the ballots going up multiple elevators and racks. And I think a lot of well-meaning people and a lot of honest people were there doing that. They were trying to participate in the process. And I would say that 99% of the people there, the way the process was designed, I believe that people thought it was a non-fraudulent process.
Gregory Stenstrom: (01:02:34)
I heard that said many times. I said, “There’s no fraud going on here.” I said, “Well, I’m not…” I didn’t even bring that up. But I think people saw what they wanted to see, and they saw what was intended for them to see. I called it, at one point, Kabuki theater. I said, “It was all designed for us to see it. It was entertaining. There were cameras on it.” When we finally got into the back room where the votes were being ingested, as a data scientist, I want to see where the data’s coming in. And I wanted to know the universe of the votes. Well, the universe of the votes was only supposed to be 120,000 mail-in ballots. We were told there were 6,000 ballots remaining. So, I said, “Okay, we have a universe of 126,000 votes.” And then, when I get back there, the universe wasn’t 126,000 votes. The universe was 200,000 votes. So, that’s a problem.
Gregory Stenstrom: (01:03:28)
A couple of other things is, the BlueCrest sorter machine was only manned by one person. People ask me all the time, “How do people commit crimes?” I know there’s a lot of theories here. And I always look for the simplest thing. People are sticking USB sticks in, putting ballots in, very simple thing. Only takes a couple of people. It doesn’t take a big conspiracy. I think people look at things, and they use inflammatory words, like fraud and so forth. As a forensic computer scientist, my interest is in the data. Where did it go? Where…
Gregory Stenstrom: (01:04:03)
My interest is in the data. Where did it go? Where did the spoilers go? How did the data come in and go out of the system? So I think as a scientist we need to look at that and we need to audit that. What was really upsetting to me, most upsetting, was I had spoke to multiple law enforcement agencies and literally begged multiple law enforcement agencies, I said, “Go in.” And I said in order to prove that nothing’s happening and either exonerate yourselves from the process or refute what I’m saying, please, it’s a very simple process, just go get the forensic evidence from the computers. It’s a simple process, you turn the computer off, it’s nondestructive, takes moments, maybe half an hour, 20 minutes, to do it properly and collect the evidence. You open the computer up, you take a device called a bit blocker, you put it in the hard drive, it’s done under the observation of law enforcement officers, you take a forensic image of the drive, put it all back together, it wouldn’t have taken more than an hour to image all five machines. That was never done, despite my objections and that was three weeks ago.
Gregory Stenstrom: (01:05:15)
Lastly, when they said, “Well we’ve got all the forensic records and so forth,” we just learned two days ago that virtual all chain of custody logs, records, yellow sheets, everything, was gone. All forensic evidence, all custody sheets in [inaudible 01:05:39] County are gone. They had a signing party where they sat down and poll workers were invited back to recreate those logs and our understanding is as of today, was that they were unsuccessful in getting them all. So we have a situation in where we have 100,000 to 120,000 ballots, both mail in and USB, that are in question. Now there’s no cure for this, there’s no remedy for this. As a home charter we could have a re-election in Delaware County for our own representatives within our own town. But there is no cure for that for the president of the United States. And I don’t believe, as a citizen and an observer to this, that anybody could certify that vote in any good conscience.
Gregory Stenstrom: (01:06:33)
And if the democrats, that have a part of this process, had done things, followed their own procedures, which they created almost unilaterally, we would be in a situation where they could exonerate themselves and they could say, “Mr. [inaudible 01:06:49], you’ve been misinformed. We have evidence here that refutes what you say.” But that’s not the case. They can’t do that. So I say if you can’t certify that vote, and you can’t certify 100,000 votes out of 300,000, then you can’t certify Delaware County. And I’m done and thank you for your time and patience.
Speaker 4: (01:07:11)
Thank you all to this panel. Before I have Senator Mastriano introduce the next 15 minute panel, I want to note that we’ve also been joined on Zoom by Representative Kathy Rap from Northwestern Pennsylvania. Doug?
Doug Mastriano: (01:07:31)
Thank you, panelists. If you can stay in the area we’re going to bring up the next group here. We’re going to save questions and answers for the end. Mr. Mayor, I understand my list might not be synchronized. I’m going to ask you to introduce the next folks coming up to testify. If you could pass the mic to the mayor, somebody. Thank you all.
Rudy Giuliani: (01:07:51)
We could call up four more witnesses like this or we could now put on the statistical expert who could explain some of this and then put on the additional witnesses. Either way you prefer. Okay then we’ll call Colonel Waldron. And if you don’t mind, if I can interrupt and ask him a few questions, can probably get it directed. It’s good for me to do that because I don’t understand it. I’m only kidding. Colonel, please introduce yourself.
Phil Waldron: (01:08:46)
Good afternoon Senator, Colonel and gentlemen, ladies. My name is Phil Waldron. I’m a retired Army Colonel, 30 years. I spent the first half of my career just like Colonel Mastriano here as a calvary officer conducting armed reconnaissance, counter reconnaissance. Last half of my career spent in information warfare as a psychological operations officer and information operations officer, conducted computer network operations, electronic warfare, special electronic warfare, deception, counter deception and op sec and a couple of other specialties.
Phil Waldron: (01:09:28)
[inaudible 01:09:28] He’s still got stets in his purse. So I spent all of my time as an operations officer. My perspective looking at this problem set is how to break in, how to break it, how to destroy it, how to manipulate it. And my team has been researching this specific issue since August but we’re working with another team that’s been intently working on this problem set, the voting machine manipulation, for two years, since the Ted Cruz and Beto race in Texas in 2018 and the Kentucky governor’s race where there was significant anomalies observed. And I like to add that there are many, many more teams like ours. Small teams that are joined in this fight and they’re throwing the flag left and right so there’s a lot of folks who are recognizing anomalies.
Phil Waldron: (01:10:24)
The voting systems in the US and in Pennsylvania were built to be manipulated. They’ve been used in elections around the world, in stolen elections around the world, in Venezuela, Italy, Argentina, Singapore, Bolivia as close as two weeks ago. Philadelphia uses ESNS, Pittsburgh uses Dominion, other counties in Pennsylvania use Dominion and other systems so what’s the real deal? All these election systems have a common DNA. SGO Smartmatic sold Sequoia voting systems to Dominion in 2010 and then the Debold company spun off Premier Election systems to Dominion as the result of an anti-trust suit. So the bottom line is that these systems have similar code and similar functions. And just so you know, I know there have been statements to the contrary but I personally debriefed the son of a Cuban intelligence officer who had first hand knowledge of Hugo Chavez’s family members who told him not to worry about the populous threat against Moduro’s election in Venezuela. “That it was guaranteed. Their father invested the money to build the SGO voting machine system.” So I have no reason to doubt this gentleman, he’s sworn an affidavit to this effect. But that’s the root of the SGO voting machines.
Phil Waldron: (01:11:49)
So these systems are not what you’ve been told. They are connected to the internet and servers outside of the US. They’re connected from the top to the bottom and the middle. There is no transparency, as our previous witnesses mentioned, as to how the voter information is processed, how and where it’s stored. The voting record is able to be modified and/or deleted by operators, administrators and outside threats. Operators can assign votes for write in ballots, blank ballots or error ballots in large numbers so that they can be directed toward one candidate or another at the operator’s or supervisor’s discretion. Many experts have published how easily these machines can be hacked to manipulate votes. As a matter of fact, one of our white hat hackers previously discovered a malware that’s present on the servers that captures every log in and every password of every operator down to the precinct level that logs into one of these systems. That’s just like giving the password to your bank account out, putting it on the dark web. It’s not going to be there very long.
Phil Waldron: (01:12:58)
And just so you probably all are aware, on 30 September, an election storage facility was robbed in your state. 30 USB devices were stolen and a laptop. Those USB devices more than likely had encryption devices and you just heard another previous witness talk about the nonstandard use of the USB storage devices.
Phil Waldron: (01:13:25)
So these systems, in a nutshell, allow authorized and unauthorized users to cancel votes, shift votes, preload votes, vote blank ballots, all in real time and in large numbers. They’re connected from the top to the bottom. So one bad actor or a team of bad actors can have equally negative influence. It’s been described by a person in another state that we’re working with, just like the lotto. Whoever organizes the lotto is always going to win. It’s controlling the numbers and it’s controlling the margins. Our experts and other academics believe that up to 1.2 million Pennsylvania votes could have been altered or fraudulent. This is what we discovered in the last 22 days. Really only a detailed forensic analysis of the actual machines and software will truly show how many Pennsylvania citizens have had their civil rights violated.
Phil Waldron: (01:14:24)
So to use these type of machines with little or no audit trails, little or no transparency of how the votes are processed, where they go, where they’re stored, will never leave the public satisfied that we truly have a representative democracy. I’d like to correct something the mayor said, I am not a statistician. I’m a combat officer and didn’t do well in math. But I can understand the numbers that he put out earlier.
Phil Waldron: (01:14:55)
I want to share with you one chart, I believe it’s in your package. So what our team has done is focused on the spike anomalies. And these are events where a numerical amount of votes are processed in a time period that’s not feasible or mechanically possible under normal circumstances. I believe Greg Stinstrom mentioned earlier, the processing of these ballots through machines, there’s a manufacturer specified rate of speed that a number of ballots can be imaged and processed. These spike anomalies in this chart really show where for us to look forensically to actually determine what happened with these votes. Our team has looked at these systems and there are a dozen ways to interdict the voting process, whether it’s mail in ballot manipulations, they can scan and allocate blank votes, whether it was a 70,000 votes left in the back room. There’s just lots of ways to interdict these systems.
Phil Waldron: (01:16:11)
So with that I’ll answer any potential questions.
Doug Mastriano: (01:16:14)
Thank you, next panel please.
Rudy Giuliani: (01:16:16)
Can I just ask him to clarify one or two things? When we look at this Pennsylvania fixing the vote chart that they all have, could you explain at the very beginning what that line means, Biden injection?
Phil Waldron: (01:16:37)
so at the very beginning of the chart where there’s a circle it says on election day, what that indicates is there is a spike in loaded votes, 337,000 plus or minus some votes that were added in there in one big batch. So that was an anomaly in the reporting. Normally you would expect to see a smooth curve going up, not any big spikes. That’s kind of what Greg was talking about, the anomalies of loading and uploading those votes. So that big spike that occurs there is a prime indicator of fraudulent voting.
Rudy Giuliani: (01:17:20)
And that’s 604,000 votes in 90 minutes, is that right?
Phil Waldron: (01:17:25)
Correct. This is 337,000 votes.
Rudy Giuliani: (01:17:29)
In that period of time?
Phil Waldron: (01:17:33)
Yes.
Rudy Giuliani: (01:17:34)
And when you look at this entire curve, with all these spikes, can you calculate how much of a vote that accounted for for Biden and how much for Trump?
Phil Waldron: (01:17:46)
Close to 600,000. I think our figures were about 570-some odd thousand that all those spikes represented over time.
Rudy Giuliani: (01:17:53)
For Biden?
Phil Waldron: (01:17:54)
Correct.
Rudy Giuliani: (01:17:54)
And how much for Trump?
Phil Waldron: (01:17:56)
I think it was a little over 3,200.
Rudy Giuliani: (01:18:04)
Now, just to go back to your original document, this one pager that they all have. Mail in ballots counted without being observed, those were the ballots we were talking about that were not observed in Alleghany County and in Philadelphia, is that right?
Phil Waldron: (01:18:24)
Correct.
Rudy Giuliani: (01:18:24)
682,770. Now this is the part that is a mystery. Mailed ballots sent out, 1,823,148. But when you go to the count of the final count of the vote, there were 2,589,242 mail in ballots. What happened? How do you account for the 700,000 mail in ballots that appeared from nowhere?
Phil Waldron: (01:19:01)
So our cyber team uses white hat hacking techniques. They gather a lot of publicly available information and that information was from the Secretary of State’s website. That website has been updated as late as 11:16 this morning with provisional mail in ballots so those numbers are still changing. They changed last night so it’s a continual target.
Rudy Giuliani: (01:19:29)
It’s 22 days after the election.
Phil Waldron: (01:19:31)
That number, the 2.5 million number is no longer on the website.
Rudy Giuliani: (01:19:39)
It’s just been taken off?
Phil Waldron: (01:19:41)
It’s not there anymore.
Rudy Giuliani: (01:19:42)
Is there any explanation for why it’s been taken off?
Phil Waldron: (01:19:44)
There is no indications.
Rudy Giuliani: (01:19:46)
But has there been a change made in the 2,589,242 mail in votes that have been counted in the total vote.
Phil Waldron: (01:19:58)
I’d have to check the Secretary of State’s website as of after 11:15.
Rudy Giuliani: (01:20:00)
And could you also check and see is there any change in the 1,823,148 ballots that were sent out?
Phil Waldron: (01:20:10)
The mailed out ballots number seems to be holding steady.
Rudy Giuliani: (01:20:14)
And was there any other method of producing ballots other than sending them out?
Phil Waldron: (01:20:21)
Not that we’re aware of unless, as previous witnesses has testified, the potential for multiple ballot counts. So the ballots could have been counted twice.
Rudy Giuliani: (01:20:31)
And have you ever gotten the chance to examine any of these ballots?
Phil Waldron: (01:20:34)
No. That would be part of the forensic process. One suggestion, whoever does the analysis, is using paper and ink analysis of the micro photo spectrometer. That would analyze the ink on those ballots to see if they were mass produced.
Rudy Giuliani: (01:20:53)
So in addition to the 682,770 ballots that were entered without a single inspection of any kind, there also appear to be something like 700,000 mail in ballots that were never sent out that were counted.
Phil Waldron: (01:21:14)
There are noted discrepancies.
Rudy Giuliani: (01:21:17)
That’s a pretty big discrepancy.
Phil Waldron: (01:21:19)
Yes, sir.
Rudy Giuliani: (01:21:19)
And that’s been there.
Doug Mastriano: (01:21:22)
Thank you. We do have to press on to the next panel since we have a lot of questions we’re attending here. Mr. Mayor, could you introduce the next panelist?
Rudy Giuliani: (01:21:29)
So now we have Jane Winters, Gary Phelman, who’s going to be on Zoom. Is he here? Then have him come.
Doug Mastriano: (01:21:42)
Please come up as you’re called.
Rudy Giuliani: (01:21:44)
I believe Charles Nudo, N-U-D-O. And David Stisogis. I think I have them all. I think I have them all.
Doug Mastriano: (01:22:08)
Thank you, gentlemen. We’ll start with the men physically in the room here with your testimony then go to the Zoom after that. So please.
Gary Phelman: (01:22:15)
Thank you, Senator. My name is Gary Phelman. I am a resident and voter of Philadelphia City and County. I’m registered as a republican. I have been involved with [inaudible 01:22:27] republican politics for over 20 years now. I’ve had watcher certificates and on this past November 3, 2020, I was issued a watcher certificate to be the watcher for President Donald Trump. This is the watcher certificate that I was given and it is issued for President Donald Trump to me.
Gary Phelman: (01:22:58)
These watcher certificates, I’m not sure the exact year, I’m going to say about 2002 or 2003, are now for county wide. So everywhere in the county the watcher certificate is good for. So [inaudible 01:23:14], is city and county so this watcher certificate which reads, “This certificate authorized the individual to watch in any ward or division in Philadelphia.” And that’s the top line. If you want to see the watch certificate I’ll gladly bring it up.
Gary Phelman: (01:23:32)
So anyway, I was in my vehicle as volunteering for the president with an attorney and he was monitoring any kind of problems that could happen in Philadelphia. His name was Will Chamberlain and he was pretty good on Twitter and other social media stuff. So we got a word that they were not letting republican watchers in the poling places in South Philly. So we went to a place, which happened to be a funeral parlor, where they were doing the election and I walked in with the watcher’s certificate in both hands to make sure they wouldn’t snag it out of my hand. They immediately wanted me to identify myself. One of the problems that happens is republican watcher certificates are this gold/orangey color where the democrats are a light blue. I’ve never really seen one but I’ve heard they’re light blue. So they knew right away that we’re republicans coming in there to watch.
Gary Phelman: (01:24:47)
So two poll workers, I don’t know who they were, they were wearing masks and everything, I produced the certificate and she started saying, “It’s not good here.” And I’m like it is. And I said, “Please read the top line.” The gentleman that was with her said, “Would you like to go outside and talk about this?” I was not looking for a confrontation. And the other thing is too, I did have a bandana on, they had their face shield but they weren’t practice social distancing. They were right on top of me. So we did step outside, because he wanted to step outside. Will Chamberlain is not a Philadelphia resident so he’s not offered a Philadelphia watcher’s certificate. And he proceeded to take a video of me having a confrontation on the street with these two … I don’t know who they were. I don’t know if they were poll workers, they never identified who they were.
Gary Phelman: (01:25:59)
And the video, which was released to the public, is a little over 30 seconds on Twitter and it already has 3.8 million hits on Will’s Twitter page. There is a complete video but it’s on my phone and I made it available to the Senators, it’s over a minute, about what happened on the street. I tried to explain this watcher certificate is for the city of Philadelphia, you’re in the city of Philadelphia and you have to let me observe. I’m the eyes and ears of the president of the United States and they wouldn’t allow that.
Dave Stisogis: (01:26:45)
Thank you for the opportunity to be here today. Oh sure.
Gary Phelman: (01:26:50)
I just want to add another thing that’s really kind of crucial. When this happened, the fact checkers group or organization, I don’t even know what they are, started to put disclaimers on the video. Fact checkers said they contact me, fact checkers never contacted me. Fact checkers said that it was a misunderstanding, they said the commissioner’s office tried contacting me, they used the name Kevin Feely from the commissioner’s office. I don’t know who Kevin Feely is, nobody from fact checkers called me, I don’t even know if they have my phone number. And the commissioner’s office did nothing to follow up on any of this. I did see commissioner Al Schmidt when we were doing the canvasing but we didn’t mention anything about what happened that day.
Dave Stisogis: (01:27:50)
Good afternoon. I’m Dave Stisogis. I’m an attorney, licensed to practice law in Illinois and Florida. Some time ago I was a Cook County Assistant State Attorney prosecutor and during that time I actually did election law enforcement during the course of that time, including seeing to it that people had appropriate access, precinct openings and closings, that machines were zeroed out. Since that time I’ve either been a candidate or a campaign in over 10 other individual elections and in 2016 I actually worked as a volunteer for the Trump/Rubio campaign in Broward County, Florida. I discovered them opening mail in ballots in a locked rom. Before that was all said and done we actually had a Broward County judge monitoring everybody’s access. He was actually on site monitoring everybody’s access to see to it that that couldn’t happen, see to it that everybody would have appropriate access.
Dave Stisogis: (01:28:50)
Currently I am lead counsel in conjunction with Judicial Watch in a suit called Illinois Conservative Union versus the Illinois State Board of Elections for violations of the board in terms of the National Voter Registration Act. Given that kind of background will give you some kind of idea of where I’m coming from on this subject.
Dave Stisogis: (01:29:13)
On October 24 this year I came to Pittsburgh as a volunteer with Lawyers for Trump and I was asked to organize folks to do monitoring of the pre-canvas activities during the course of the next week. Myself with some other volunteers made over 500 phone calls, we recruited 50 individuals that were willing to come and canvas or come and monitor the pre-canvas along with 24 other out of state lawyer volunteers.
Dave Stisogis: (01:29:47)
We had organized all this because of course we had 19 in Alleghany County there were 19 republicans on the ballot and so we were able to have credentials through the RCAC for 19 watchers at any given moment. So we had to organize them. So we organized everybody during the course of time and they had given us credentials in advance and then two days before the election, the Friday before the election, we got a call from the electoral board and said, “Oh by the way you need to submit everybody’s credentials in advance. And you need to have everybody’s signatures on the credentials that you signed in advance.” And of course we already had put together a team of 70 and we’d allocated them four hours of time in four hour shifts to be able to monitor through the course of the whole period of time and then over the next three days we scrambled like heck to try and get signatures from the various volunteers and other counsel that were assisting us.
Dave Stisogis: (01:30:52)
Needless to say, we lost about 20-25 folks that we were unable to get the credentials to in advance. And get the signatures back and forth and all the things necessary to do that. So I wanted to mention, you’re hearing about huge global issues but it goes down to the petty as well. It goes down to the petty as well in terms of interfering with his access and interfering with changing rules. I know Secretary [inaudible 01:31:23] changed some huge rules but the local boards, day in and day out, were changing rules as well to make the monitoring process next to impossible.
Dave Stisogis: (01:31:34)
At any rate, we did that and then we wound up, I went with a group at about 6:45 on election day to 901 Pennsylvania Avenue in Alleghany County where they were doing the pre-canvas. And at the time when we arrived we were herded in to what is best known as the corral. I’m sure you’ve seen or heard about those things. It was a fenced in area, the fence was maybe two and a half, three feet high. Closest anybody could get to make any observations was maybe 15-20 feet from the closest ballot processor to maybe 150 feet, because we were in maybe a 20,000 square foot area where the biggest activity was taking place. But I would mention that the mail in ballots were coming into that room already sliced open at the top. We have no idea when or where the slicing took place, where the opening took place or what was in fact inside those mail in envelopes.
Dave Stisogis: (01:32:45)
At that point in time folks were then separating the mail in ballot from the secrecy envelope. You could see that’s exactly what they were doing, they would put one on this side and one on the other side. And then they’d take them and put them some place that I have no idea and then the secrecy envelopes would go to another slicing machine and that’d be sliced open and at that point, then, the ballots would come back and they’d be divided from the secrecy envelopes.
Dave Stisogis: (01:33:11)
We had really no concept of what was going on. In the room next door, there was a smaller room around the corner that you had to leave the 20,000 square foot room. There was one about 7,500 square feet and in that room, there was a machine and the machine would be, apparently, sorting things by the barcodes and then eventually, somehow or other, people would come in with big armloads of ballots from the other room. Apparently with no providence, no explanation of where they came from, and here’s an interesting thing that you guys might consider when you’re doing this in the future, one of the biggest bottlenecks would take place at this point in time because all these ballots came in tri folded. And all the ballots had to be flattened. And so they would take the ballots and put them under big stacks of reams and reams of paper to flatten them out and then there’d be workers kneading the ballots to try and make them so that they could go through the machines.
Dave Stisogis: (01:34:19)
And when everybody’s saying you haven’t counted anything for X number of hours, they said, “We’re trying to flatten the ballots.” It was absurd and obscene. So at any rate, when everybody was waiting, that’s what was going on, they’re flattening the ballots for the most part. And then there was about six machines where they were feeding the ballots and while they had six machines feeding the ballots, as they would go in, two or three would kick out to a different tray than the other tray. And workers sometimes would take the ones that weren’t kicked out and put them in a stack and then sometimes they’d run all of them through again and sometimes run two or three. We never had any idea if they were zeroing out a machine, what they were doing. I asked the county solicitor who was there, I said, “What’s going on?” He told me that this was a Pitney Bowes representative working there that had been sworn in as a county representative and that sometimes the ballot machine doesn’t count those things. So he runs them through.
Dave Stisogis: (01:35:29)
But there was absolutely no providence to what was going on in the counting machines. I have no idea because of course they would just take stacks of ballots that had been sitting under reams of paper trying to be flattened and then run them through the machine. So we have no idea about that. And so at one point I asked the county solicitor, I said, “Can you describe for me the journey of a ballot from the time it hits the door to the time that it’s counted? Where does it go? What’s the process, et cetera?” I-
Dave Stisogis: (01:36:01)
Where does it go? What’s the process, et cetera? I got a short answer. “No.” I then asked him, I said, “Can you give me any written protocol about what is supposed to happen? It hits the door. Then it goes through a machine, theoretically sorted by barcodes, from what I understood from someone else so that if there’s a challenge in advance of the election, they know where to look,” because, of course, at five PM on the Friday before elections, you can challenge things for people having died and a few other reasons.
Dave Stisogis: (01:36:37)
But then I asked, I said, “Is there any written protocol for that?” He said, “No, we have no written protocol for what we’re conducting, what we’re doing here.” So he answered. He’s supposed to be the one giving legal advice to people on how to legally handle ballots, and he tells me that he doesn’t know how they’re handled and he tells me that there is no written protocol on how they are supposed to be handled.
Dave Stisogis: (01:37:05)
So I might suggest that there was just literally no way to change. I mean, there was no way to challenge. There was no way to look at those ballots, and I would say in my experience, electoral experience, one of the things that legitimizes elections is the ability of not a disinterested third party to review what the process is, but actually an adversarial third party to review what the process is. This was completely and utterly absent.
Dave Stisogis: (01:37:40)
I had about 25 other affidavits from other folks that had joined me during the time that had been part of this that describe essentially the same thing over there in Allegheny County, most of whom were attorneys and had been versed on the comings and goings of the Pennsylvania Supreme Court, which I might suggest, ultimately, the last decision in recanvassing when the Pennsylvania Supreme Court ultimately said that it doesn’t matter how far you’re away from the ballots because you don’t have the right to challenge anything, anyway. I read the opinion, and I said to myself, “The Pennsylvania Supreme Court just called off elections in the state of Pennsylvania.”
Dave Stisogis: (01:38:34)
I would suggest that the remedy for what the Pennsylvania Supreme Court does lies with the distinguished legislators that are before me. But the fact is is if there is no opportunity for anybody to look at them and the court has the temerity to say, “It doesn’t matter. It doesn’t matter if you come in or not. It doesn’t matter how far you are, because even if you’re there, you don’t have any right to do anything.”
Senator Doug Mastriano: (01:39:03)
If you could summarize, sir, we are running [crosstalk 00:03:07].
Dave Stisogis: (01:39:07)
Yes, I’m sorry. I’m sorry. Yeah, I apologize for being an attorney. No, I apologize, Senator. Yeah, I’ll summarize in one thing. One of the guarantees in the United States Constitution is that, in fact, every state shall have a republican form of government. I would suggest to you if the Supreme Court of Pennsylvania determines that they’re going to run the state and allow the local election authorities to essentially appoint winners, the State of Pennsylvania no longer meets that guarantee as a republican form of government. Thank you.
Senator Doug Mastriano: (01:39:48)
Thank you, sir. If we could go to our two witnesses on Zoom now, if you can come forth. Go ahead and start when you’re ready.
Elizabeth Preate Havey: (01:40:06)
Hi. My name is Elizabeth Preate Havey. I am an attorney and a resident of Montgomery County and the chair of the Montgomery County Republican Committee. Thank you, senators and Mayor Giuliani for providing me the opportunity to speak.
Elizabeth Preate Havey: (01:40:22)
After the primary, our committee prepared and provided to the PA legislative leaders and members of the Montgomery County staff in charge of the election a 30-page report of the mail-in ballot and Election Day problems and irregularities we experienced in Montgomery County. We made detailed recommendations for changes with the hope that the general election would be smoother. Unfortunately, that was not the case. There are a number of issues that evidence a total lack of transparency regarding the handling of the general election in Montgomery County.
Elizabeth Preate Havey: (01:40:56)
First, the Montgomery County Republican Committee was not provided meaningful view of the mail-in ballots at any time, despite our requests. Second, we were not provided with regular, detailed information about the mail-in ballots over the course of the election, despite our requests. Third, we still lack complete and detailed information about these ballots despite our written requests.
Elizabeth Preate Havey: (01:41:20)
We were provided our first opportunity to view and learn about the canvassing site on Sunday, November 1st, just two days prior to the election. At that visit, the chief operating officer of the county showed us the room where the canvass was to take place. To our shock, we were told we could not directly check the mail-in ballots. After some significant pushback by me, the county agreed to allow two watchers inside the room, but they were never able to see the ballots up close.
Elizabeth Preate Havey: (01:41:50)
Again, there was no meaningful observation of the ballots and absolutely no check of the ballots by anyone other than the county staff, which is all run by the Democrats, as to whether or not these ballots met election code criteria. At the most basic level, we were not even able to check to see if a voter voted twice or if a voter was deceased. With the removal of a signature being a check for existing voters, there was no way to protect against someone fraudulently voting for a registered voter.
Elizabeth Preate Havey: (01:42:21)
In addition to having no meaningful observation or review of over 200,000 mail-in ballots, we were not provided with the change in rules by the county to allow the curing of mail-in ballots in a timely matter. On Thursday, October 29th, I received an email from Frank Dean, who ran the election for the county, with the list of ballots with what they described as defects and if the voter had been contacted by the county to cure the defect. At the tour of the canvass on November 1, we were shown bundled mail-in ballots divided by precinct which the county identified as having a defect and explained that those individuals had not cured these ballots as of November 1st. The county did not provide the Montgomery County Republican Committee with an opportunity to have an authorized representative present in what was effectively the pre-canvass [inaudible 01:43:12] pre-canvass of the ballots when it reviewed and contacted voters about their defective mail-in ballots.
Elizabeth Preate Havey: (01:43:19)
After immediate investigation, it was clear that other counties across Pennsylvania were not allowing this illegal pre-canvassing to take place. In particular, our neighboring county, Berks County, a county that the president won by over 8%, interpreted the law as written and did not contact voters to cure ballots weeks prior to the election, as Montgomery County did. A portion of Berks County and most of Montgomery County share one congressional seat. So voters were not treated equally in these two counties in the same district.
Elizabeth Preate Havey: (01:43:48)
Since the election, we have received many calls and emails from Republicans with questions about whether their mail-in ballot was counted, expressing concerns that they didn’t request a mail-in ballot, but received one, anyway, that they were made to vote provisional when they shouldn’t have, that they have great concerns about the efficacy of this election. There is a real disenfranchisement of voters across Montgomery County. Many people feel that their vote did not count. Because we lack the information we need to tell them otherwise and because we were not able to have any meaningful check on mail-in ballots, we cannot provide these concerned voters with the assurance they need that the process was fair and that the election was honest.
Elizabeth Preate Havey: (01:44:31)
In addition to the lack of transparency we experienced in Montgomery County, the Kathy Barnette for Congress campaign found with just the minimal information that we’ve been able to obtain that at least 188 deceased people in Montgomery County voted in 2020. They also found concerning anomalies in the results. For example, more people voted in the small town of Norristown for the very first time than the voters who vote regularly. This occurrence is far outside the norm of what we have seen in past elections. These numbers must be reviewed and analyzed. In fact, a full review of the mail-in ballots across the state must be done.
Elizabeth Preate Havey: (01:45:15)
In order for this country to have trust in the electoral process, elections must be viewed as open and transparent. Both the Democrats and the Republicans should be provided with the same information throughout the process, and there should be meaningful opportunity to observe and object, not last-minute disclosures and rule changes. This legislature must reform the election law to provide for such transparency all along the mail-in ballot process from start to finish, and people need to feel confident that the same rules apply to each and every voter in every town and in every county in this state. Thank you.
Senator Doug Mastriano: (01:45:54)
Okay. Next member on the Zoom, you’re up. Thank you. Do we have contact?
Speaker 5: (01:46:06)
She’s still on.
Senator Doug Mastriano: (01:46:07)
Okay. There you are.
Speaker 6: (01:46:08)
We’re ready.
Senator Doug Mastriano: (01:46:09)
Please proceed.
Julia Vahey: (01:46:10)
Julia Vahey.
Speaker 5: (01:46:12)
Oh. She’s on.
Senator Doug Mastriano: (01:46:13)
Go ahead.
Julia Vahey: (01:46:15)
Hello. Thank you for having me here today. My name is Julia Vahey, and I’m a resident of Montgomery County and the Executive Director of the Montgomery County Republican Committee. I’m here to speak with you today regarding my experience as an observer of the pre-canvass and canvass of the mail-in ballots. In Montgomery County, we had over 250,000 mail-in ballots requested for the general election. In the weeks leading up to the election, I personally trained over 100 volunteers to inspect and monitor the counting of these ballots to ensure the legitimacy of the vote in Montgomery County. Unfortunately, they were never given this opportunity.
Julia Vahey: (01:46:48)
On Sunday, November 1st, during a walkthrough of the mail-in ballot facility, we learned that no watchers were permitted to inspect or even see the outside envelope of the ballot. After some significant pushback by our chairman, Liz Havey, the county agreed to allow two watchers inside the room, of which I was one. As an observer at the canvass, I was corralled into a small, narrow holding area, which is blocked off by tables and plexiglass barriers in the corner of the room. Due to the room layout and location of the holding area within the room, I only had visibility to the space where ballots were flattened and scanned. This was a huge area, and the closest scanner was roughly six feet away and the farthest approximately 25 yards away.
Julia Vahey: (01:47:31)
From my position within the holding area, I had no visibility or access to the room where ballot were sorted, outside envelopes were inspected, and both outside and secrecy envelopes were opened. The space was divided from the scanning room by a large white wall, blocking all visibility to the slicing machines, the BlueCrest sorter, and the ballot storage room. In my opinion, this was a totally separate room and not one room, as the county election board argued.
Julia Vahey: (01:47:58)
During our walkthrough of the campus facility on Sunday, November 1st, we argued that this was a separate room and was told by the county chief operating officer that we would be able to view this space by the security camera footage. The footage from these cameras was streamed to televisions in a conference room on the other end of the building, which was approximately a four-minute walk away from where the ballot canvass room was. This system was not set up on our walkthrough day, and it was not until I arrived back at the facility at seven AM on Election Day that I could see the footage was extremely poor quality and that you could only see pixelated images of workers moving around the room and had no meaningful visibility of the ballots.
Julia Vahey: (01:48:36)
Over the last ten months, in my role, I’ve spoken to thousands of voters firsthand who have lost faith in the election processes and procedures in Montgomery County and across Pennsylvania as a whole. Today, I still lack the information needed to properly inform voters that their vote was in fact counted. I thank you for holding these hearings today and hope that the appropriate legislative changes are made to restore confidence in our elections. Thank you.
Senator Doug Mastriano: (01:49:01)
Thank you. Facts are stubborn things, and I appreciate you guys laying it out there. It does make a difference. Mayor, if you can introduce the final panelists before we go to question and answer time.
Rudy Giuliani: (01:49:14)
I believe there are two other-
Senator Doug Mastriano: (01:49:17)
Two more on Zoom?
Rudy Giuliani: (01:49:18)
… Zoom.
Senator Doug Mastriano: (01:49:18)
Okay. Please come forth, friends on Zoom, when you’re ready.
Charles Nudo: (01:49:24)
Here we are. We’re here, but we can’t see ourselves. Can you hear us?
Speaker 7: (01:49:30)
We hear you loud and clear. Please begin.
Charles Nudo: (01:49:36)
Okay. My name is Charles Nudo. I live in Drums, Pennsylvania. Can you hear me?
Speaker 5: (01:49:43)
Yes.
Charles Nudo: (01:49:45)
Can you see me on the screen?
Barbara Sulitka: (01:49:46)
You’re never on the screen.
Speaker 7: (01:49:49)
No, but please continue.
Charles Nudo: (01:49:54)
Okay. Go ahead. My mother … No, we got somebody else.
Senator Doug Mastriano: (01:50:04)
Well, let’s go with what who got. Please start.
Charles Nudo: (01:50:05)
Oh. Can you see us?
Speaker 5: (01:50:08)
We see two ladies.
Senator Doug Mastriano: (01:50:09)
Yes.
Charles Nudo: (01:50:09)
Can you see us?
Senator Doug Mastriano: (01:50:10)
Yes. We have the two ladies on the screen.
Speaker 5: (01:50:11)
[crosstalk 00:14:10].
Cheryl Nudo: (01:50:12)
Barbara Sulitka.
Barbara Sulitka: (01:50:13)
My name is Barbara Sulitka. I went to the voting polls on November the 3rd to vote in person. I waited two hours in line, and when I went in, I showed identification. I was taken up to the polling place. The polling man showed me what to do, turned it on, and said, “Go ahead.” I voted, and I printed it out. When I got it and I looked at it, Trump was not on there. I voted for Trump, and it wasn’t there. My daughter, I called her over and I said to her, “Look, his vote is gone. I voted for him.” So I called a pollster over, and he said, “It’s not supposed to be seen. It’s for privacy. Just put it through the scanner, and you’re done.” It bothered me. It’s been bothering me since, because I voted, and was it counted or was it flicked? I don’t know.
Cheryl Nudo: (01:51:26)
I’m speaking on my mom’s behalf also as a witness. My name is Cheryl Nudo. I was there that day. I drove her to the polls. When she got done voting, she showed me the printout, and she was saying how the president’s name that she voted for was not printed out, when all the other delegates that she had voted for were on the printout. Me not knowing, because I had not voted yet, how the printout was supposed to look, we went to the pollster that was there and asked him why the president and vice president’s name did not print out when all the other names of the candidates printed out that my mother had voted for.
Cheryl Nudo: (01:52:05)
It seemed very strange, but not knowing any better, we asked, “Well, what is the next step?” He said … Oh, I’m sorry. He said that it did not print out because that’s for voters’ privacy, that none of them print out. So we asked him what the next step was, and he said to put it through the scanner. So that’s what we did, and we left. It seemed very odd.
Cheryl Nudo: (01:52:29)
Then I went to where I live in Drums, Pennsylvania, which is about ten miles south of where my mother voted, and I voted, no problem. Everything printed out just as it should, and I scanned it through. So it just seemed very odd, and we wanted to report that, that in my mother’s area, which was Fairview Township, it seemed very odd that it did not print out. Thank you.
Charles Nudo: (01:53:02)
Okay. I’m Charles Nudo, and by the way, my mother-in-law’s 84 years old. She forgot to mention that. Anyway, my wife then …
Cheryl Nudo: (01:53:13)
She’s 80.
Charles Nudo: (01:53:14)
I’m sorry. She’s 80. My wife called me that day, and she was distraught and told me everything they just told you. So I go to my polling place around 3:30, 4:00 here in Drums, and when I first walked in, a young gentleman came up to me, started explaining how to vote. He said, “Take the stylus, make your choices, and then print it out with a printer that’s underneath the machine.” I thought, “That’s odd.” I’d been voting since 1992. We never had to print a ballot, and I actually thought it was easier to vote in ’92 on the World War II surplus voting machines with the curtain. You did your choices, opened the curtain, and they went click, click and everything went. All of a sudden, we have to print a ballot?
Charles Nudo: (01:54:07)
So, anyway, I’m pretty good with computers. I made my choices. I printed it out. All my choices are on there, from president on down ballot, and he said, “Now you have to put it in the scanner.” He turned over a sample ballot and pointed at a weird triangle in the corner and smiled at me. I didn’t know what he meant. So I go over to the scanner, and I’m wondering, “Well, a scanner works facedown when you scan,” because I have one at home. “Do I put it in facedown or face-up?”
Charles Nudo: (01:54:39)
So he’s already across the room, talking to another gentleman that’s about 20 years older than me, trying to tell him how to do it, and I said, “Are these face-up or facedown?” He just nodded. He didn’t say either way. He just nodded yes. So I put it in face-up. So I don’t even know if it scanned the blank backside or the voting top side. Anyway, I overheard that conversation, not wittingly or that I wanted to. A man … I’m 58. He was about 78, saying … They said to him, “Now, do you understand?” He said, “Yeah, yeah, yeah, I think, but my memory’s not too good.”
Charles Nudo: (01:55:20)
When I walked out of there, I realized it’s total voter suppression. My mother-in-law here did not even get a cell phone until last year, has never sat at a computer and keyed anything in in her life, has no wifi or Internet service center house. Neither does my mother. She has a cell phone, but it’s a flip type. These are the people that went by the thousands or millions to our polls and had to go through this system, and I just wanted to point that out. That’s all I have. Thank you.
Senator Doug Mastriano: (01:55:56)
Thank you.
Speaker 7: (01:55:57)
Thank you. Senator Mastriano, before we turn to our next panel, I want to note that Senator Camera Bartolotta has joined us from Southwestern Pennsylvania on Zoom and Senator Bob Mensch from the Southeast, Doug.
Senator Doug Mastriano: (01:56:14)
Thank you, Senator. Can we go over to the final Zoom panelist? Mr. Mayor, we have more?
Rudy Giuliani: (01:56:18)
[inaudible 00:20:22].
Jenna: (01:56:23)
[inaudible 01:56:20].
Senator Doug Mastriano: (01:56:25)
Okay. Thank you, Scott.
Rudy Giuliani: (01:56:29)
I think we have a surprise guest, Mr. Chairman.
Senator Doug Mastriano: (01:56:32)
Okay.
Olivia Jane Winters: (01:56:33)
I’m sorry. Is it my turn?
Senator Doug Mastriano: (01:56:35)
Just hold off one second. Just give me a minute.
Olivia Jane Winters: (01:56:38)
Okay. It’s really hard to hear you guys.
Jenna: (01:56:39)
All right. Are we ready, Molly?
Speaker 8: (01:56:47)
Jenna?
Jenna: (01:56:47)
Yes, sir.
Speaker 8: (01:56:49)
Okay.
Jenna: (01:56:50)
Mr. Chairman, Andrew just-
Speaker 8: (01:56:51)
Let them finish their testimony. I’m watching. It’s fantastic.
Rudy Giuliani: (01:56:58)
You might as well …
Speaker 8: (01:56:58)
Let them finish their testimony.
Jenna: (01:56:59)
He wants to finish.
Rudy Giuliani: (01:57:00)
He wants to finish.
Jenna: (01:57:00)
He wants to finish.
Senator Doug Mastriano: (01:57:00)
Okay.
Jenna: (01:57:00)
All right?
Senator Doug Mastriano: (01:57:01)
Okay, please. Can you hear me on Zoom? If you can introduce yourself and proceed.
Olivia Jane Winters: (01:57:07)
Yeah. Can you hear me?
Senator Doug Mastriano: (01:57:08)
Yes. We’ve got you loud and clear.
Olivia Jane Winters: (01:57:13)
Okay. My name is Olivia Jane Winters, and I’m a professional organizer and a business management consultant. I live in Philadelphia. I actually was asked to be a minority inspector in 3615. That’s the ward and the division. I was just purely doing this to help my mom out. She’s a ward leader in a different area of the city. I’m a registered Democrat, and I just wanted to help and make sure it was a fair election. I don’t care who wins. I care that it’s a fair election.
Olivia Jane Winters: (01:57:49)
I showed up to the polls at 6:30 in the morning. Immediately, I was met with a hostile attitude from all of the people that I had to work with. That would be the majority inspector, the poll watcher, the machine operator, and a committee man, who was electioneering in the polls. He was wearing shirts and a hat and a mask for who to vote for. We are about an hour and a half in in the morning. We have a long line of people. We must have had, I don’t know, 60 people in line in the building, snaking around the building.
Olivia Jane Winters: (01:58:34)
A woman came up and said, “I’d like to turn in my mail-in ballot so that I can vote in the booth.” The girl who was clerking for me said, “Well, if you want to turn in your mail-in ballot, we’re going to need to have you fill out this form, and the majority inspector is going to need to sign the form.” The majority inspector had decided in the middle of the morning to go home and was not in the polling place for over 45 minutes. To my understanding as the minority … I’m sorry, not the majority inspector, the judge of elections. My apologies. She had gone home, and to my understanding, the judge of elections is not to leave the polling place.
Olivia Jane Winters: (01:59:22)
So she was out of the building. We needed her to sign this form. The man who was electioneering and was a poll watcher, but also a committee man confronted myself and the clerk and said, “You don’t know what you’re talking about. Anybody can sign the affidavit. Why don’t you stop trying to cause problems? Why don’t you shut up?,” started getting in my face, cursing at me, telling me that I needed to be quiet, that I didn’t know what I was talking about and I should just learn to sit down and not say anything and just let it happen.
Olivia Jane Winters: (01:59:55)
My clerk and I said, “No, we’re not going to let that happen. That’s ridiculous. It says very clearly in our training and on this sheet that an affidavit needs to be signed by the judge of elections if somebody’s going to turn in a mail-in ballot before they can go vote in the booth.” We made her wait. Finally, the judge of elections came back, not, of course, before he was threatening me. The majority inspector threatened to slap me in the face, and he told me that it was going to become a, quote unquote, racial issue. I’m not really sure why that would become a racial issue. It has nothing to do with race. It has everything to do with following procedure and making sure it’s an honest election.
Olivia Jane Winters: (02:00:42)
I felt threatened. I called my ward leader. The ward leader then called the Commissioner for Philadelphia, Al Schmidt. Al Schmidt had me call and report that to the DA, and then about an hour and a half after that, the majority inspector told me that she wanted to vote, because we had a lull in people coming to the polls. So it was pretty quiet. I looked her name up so that she could sign the book, and it said that she needed to remit her mail-in ballot or vote provisionally. She said, “Oh, I threw that in the trash.” I said, “Well, then you have to vote provisionally.” The machine operator and the same committee man/poll watcher who was electioneering that I mentioned before got up in my face again, told me that I didn’t belong there, that I needed to shut up, started cursing expletives at me again, told me that they were going to allow her to do that and that I better be quiet about it, and then they let her go into the polls and vote. She did not vote provisionally at all.
Olivia Jane Winters: (02:01:53)
She voted, and who knows? Maybe she voted twice. Not to mention it wasn’t until I had actually called our ward leader, who had called the Commissioner, who had called the DA, that they then sent representatives down to discuss this with me and defend me. I mean, I had maybe six men there, because I was being threatened in the polling place, and the committee man that I mentioned earlier, who was threatening me, then got his cousin involved, who was working the polls as a poll watcher and was also wearing electioneering gear. She got up in my face, demanded to see my credentials, then tried to refuse to give them back to me, told me that they were illegitimate minority inspector certificates. They basically told me they were going to call my council rep and he was going to come down and make me leave, just ridiculous threats.
Olivia Jane Winters: (02:02:53)
I then testified in a hearing for the City of Philadelphia for them on Election Day regarding this harassment. Meanwhile, I had mentioned that until we had the deputy sheriff come down and tell us how to actually receive a mail-in ballot and how to vote provisionally, we were not doing it correctly. The judge of elections was not doing it correctly, and I cannot stress this enough. My brother had the same role as me as a minority inspector in a different polling place, and he said they were not doing it correctly, either.
Olivia Jane Winters: (02:03:31)
I don’t think that anybody actually really knew how to receive a mail-in ballot. Most of the books that we had to get your authorized signature, some would have a Remit Ballot little box that you could click, and others didn’t have that at all. So if somebody came in and remitted a ballot, I’d almost have to like hand-write that in just to keep it as honest as possible. There was no place for that. So I don’t see really how they were keeping track of that, because you could easily do both. I mean, it happened in front of me. My majority inspector definitely voted and then went into the polls and voted again. Unless you have questions, I’m not really sure. I think that’s pretty much what happened.
Speaker 7: (02:04:22)
Thank you. Thank you very much. Senator Mastriano.
Senator Doug Mastriano: (02:04:27)
Thank you. If we can have our final Zoom … It was one or two more. If the next one can come forth. Whenever you’re ready. Okay. Are you on? Hello? Can you hear me?
Gloria Lee Snover: (02:04:52)
Hello.
Senator Doug Mastriano: (02:04:52)
Okay. Over to you. We can hear you.
Gloria Lee Snover: (02:04:59)
Am I on?
Senator Doug Mastriano: (02:05:00)
Yes. Please begin. You have the floor.
Gloria Lee Snover: (02:05:05)
This is Gloria Lee Snover. I am Chairman of the North Hampton County Republican Committee. North Hampton County is a Democrat county where Republicans win elections. I have been involved in the political process in North Hampton County for more than 25 years. I’ve worked on campaigns for Township Supervisor to President of the United States. This year with one of the most vast, complex elections I have ever had to oversee as Party County Chairman.
Gloria Lee Snover: (02:05:40)
The complexity was due to the new mail-in ballot system and ever-changing rules, the unmanned drop boxes in the general election that we did not have in the primary, also changing the fact that voters could bring their ballot to the polls and vote on the machine and many other new procedures. There was mass confusion regarding the mail-in ballot system. I fielded scores of calls from North Hampton County voters and collected incident reports from voters and poll watchers regarding the November 3rd election.
Gloria Lee Snover: (02:06:21)
The typical calls I would field were, “I didn’t ask for a mail-in ballot. Why am I on the mail-in ballot list?,” “I did not get my ballot. What should I do? It’s been weeks,” “How come my wife got her ballot and I didn’t get one? We applied on the same day,” “How can I vote on the machines if I applied for a ballot?”, “Is it safe to put my ballot in the mail?,” “My ballot says it wasn’t received, and I turned it in to the election office in person,” “My ballot says canceled. What does that mean?”, ” I voted in-person, and it still says not counted,” “I’ve been on hold with the election office 70 minutes. What do I do?”
Gloria Lee Snover: (02:07:10)
In turn, because of these questions, I had a number of phone calls and emails, correspondence with the election office and the county solicitor before Election Day, trying to make sure I was disseminating information correctly to the voters, volunteers, and the campaign. One call turned heated when the registrar threatened to turn me over to the district attorney for posting a picture of a voter ballot-harvesting a large bag of ballots at a North Hampton County drop box. It was my opinion the registrar was more concerned we exposed it rather than the fact that it was happening. The most pertinent information I have to offer this committee was my call-
Gloria Lee Snover: (02:08:03)
I have to offer this committee was my conversation with the election office regarding curing the disqualified naked ballot, which they unlawfully pre-canvased before election day. They actually told me on the phone early in October, they felt them, they held them up to the light, they separated them out in different boxes. Reflecting back on these three weeks and the experience, the major concerns regarding this election are equal protection.
Gloria Lee Snover: (02:08:38)
Were the Democrats and Republicans treated the same? Were they given the same information and opportunity to vote? Did they equally have an opportunity to cure their disqualified ballots? Not in my county. There was no ability to properly witness the canvas of the mail-in ballots or see the actual ballots. Were there signatures? The solicitor told me there is absolutely no law that allows us to see the counting or to see the ballots. To this day, we have no idea what or whose ballots were counted.
Gloria Lee Snover: (02:09:27)
Another one, very strange, there’s a pattern of reactivated old voter rolls that kept reoccurring on the SURE system. The SURE system is our Pennsylvania voter system. Scores of people that had not voted since the 1990s, more than 22 years ago, were all now reactivated and receiving mail-in ballots. People that didn’t vote for Obama or Hillary, were all of a sudden receiving mail in ballots, setting up the opportunity for fraud on a massive scale that would go undetected, because voters who never intended to vote would not know.
Gloria Lee Snover: (02:10:15)
The SURE system is not so sure. We also found in North Hampton County, more than 1500 votes where the mail-in ballot was received in the election office on a date before it was even mailed. We have a problem with this election. Thank you for allowing me to share my election experience, and I plead with you to remedy the mail-in ballot process. Thank you.
Speaker Panel: (02:10:45)
Thank you for your testimony. Our last Zoom, are you there? Can you hear me? If so, please begin. Is there one more? Nope, that’s it. Okay. I guess a Q and A.
Speaker 9: (02:11:04)
All right. Connect it.
Mr. Chairman: (02:11:09)
Let me begin now with questions from our representatives and senators. I believe probably about 30 minutes. [inaudible 02:11:18].
Speaker 9: (02:11:18)
Mr. Chairman?
Mr. Chairman: (02:11:21)
Oh, we have one additional witness.
Speaker Panel: (02:11:23)
Well, a commentator.
Mr. Chairman: (02:11:26)
Please.
Speaker 9: (02:11:27)
All right. I will introduce to you now, Mr. President, you are connected.
Donald Trump: (02:11:40)
Thank you very much. I’ve been watching the hearing on OEN, and saw a couple of other great networks, but I really appreciate being asked to speak. I’m in the Oval Office right now, and it’s very interesting to see what’s going on. This was an election that we won easily. We won it by a lot. A big energy official was on this morning, on a important show, and said, “There’s no way Trump didn’t win Pennsylvania, because the energy industry was all for him. I saw with my eyes what happened,” and he told me horror stories, absolute horror stories.
Donald Trump: (02:12:20)
This was a, very sad to say it, this election was rigged, and we can’t let that happen. We can’t let it happen for our country. This election has to be turned around, because we won Pennsylvania by a lot, and we won all of these swing states by a lot. Anybody watching television the night of the election was saying, “Wow.” I was called by the biggest political people, “Congratulations, Sir, on a big win.” All of a sudden, ballots were dumped all over the place, and a lot of horrible things happened. Everybody in that room, I want to thank all of the people that signed affidavits and all of the speakers. You’re fantastic people. You’re great patriots.
Donald Trump: (02:13:02)
I want to thank the senators for being there. It’s so important, day before Thanksgiving, it really represents somebody, between the voter suppression and all of the horrible things that happened to poll watchers. We have poll watcher affidavits piled up to the ceiling, and they’re all over. They were treated horribly all over this, in all of these swing states, I mean virtually all of the swings states. Many other things were happening that were horrible, just horrible, but the poll watchers weren’t allowed to watch. They were, in many cases, whisked out of the room.
Donald Trump: (02:13:38)
Not only into pens that were 20, 30, 40, 60, a hundred feet away, where you couldn’t even see, they were using binoculars. People are reporting that they had to use binoculars, and that didn’t work. If you were a Republican poll watcher, you were treated like a dog. The Democrats had no problem, but they were rough. They were literally pushed out, and it was rough tactics. This is what happened here, this is not the United States of America, what happened. I think everybody knows that that’s why you’re there, and that’s why you’re so vehement about it.
Donald Trump: (02:14:15)
We have many, many cases, many, many cases of people walking in. A woman, an elderly woman walks in, looking forward to voting November 3rd, and says, “Oh good. Where would I go about voting?” “I’m sorry, you’ve already voted. Your ballot is in.” I said, “No, I didn’t vote. I didn’t vote.” “No, your ballot is in. You’ve already voted,” in all cases, for Biden, by the way. She said, “No, no, I want to vote.” “Nope, your ballot is in.” Then they give her a provisional ballot to sign, which goes nowhere. It’s a disgrace that this is happening to our country.
Donald Trump: (02:14:54)
We won this election by a lot. We got 74 million votes, and if you would have said 74 million votes the day before the election, every single professional in the business would have said, “There’s no way of beating that.” We got 11 million votes more than we had four years before in 2016, and we got many votes more than Ronald Reagan had when he won 49 states. Nobody would’ve said we even had a chance of losing. All you had to do is take a look at the numbers at 10 o’clock in the evening, when everybody thought the election was virtually over, and then very weird things happened, but they’re not weird to professionals, and they’re not weird to Dominion and other people that operate machines, and they’re not weird to the people that handle the ballots where they were flooding the market.
Donald Trump: (02:15:42)
People were getting two and three and four ballots in their home. People that were dead were signing up for ballots. Not only were they coming in and putting in a ballot, but dead people were requesting ballots. They were dead for years, and they were requesting ballots. The whole world is watching us. The whole world is watching the United States of America, and we can’t let them get away with it. We have judges that are afraid to make a decision. We have judges that don’t want to do the same thing. Well, a very good good lawyer said, “Well, sir, I mean, that’s a big statement for a judge to overthrow an election.” I said, “Really? If he got hundreds of thousands of votes more than he was entitled to get, through all of the things that I’m listening to right now, and you’re just covering a few of them.”
Donald Trump: (02:16:34)
We have hundreds and hundreds of affidavits of stories that are even worse than the stories I’m hearing. Why wouldn’t they overturn an election? Certainly overturn it in your state, because we have other states that are just as bad. If you look at Michigan, with Detroit, you look at the things that happened in Detroit, where you have a voter, but you have more votes than you have voters. You take a look at Detroit, Michigan, you have more votes than you have voters.
Donald Trump: (02:17:07)
Then you have two people that don’t want to certify. They don’t want to certify, and they’re harassed violently. They turned off the cameras during the harassment for two hours. Then they said, “Wow,” and they were afraid, and they voted. Then they went back to sign and they couldn’t do it, because they said, “We can’t do it, because this is corrupt. This is horrible what’s taking place.” Think of it, more votes than you have voters, but that was the least of it. They have things that were as bad as that, and this is going on all over, all over. We’re doing very well in a lot of states. A lot of good things are happening in Georgia. We’re getting little help from government, but a lot of good things are happening in Georgia, Wisconsin, and Michigan.
Donald Trump: (02:17:51)
They’re seeing what happened in Detroit, and we sure are looking at what’s happening in Pennsylvania and Philadelphia. What happened in Philadelphia? They keep the poll watchers not only in pens, but they keep them out of the building. The only reason they got back into the building was they got a court order. Then the definition of, back into the building, was very far away where they couldn’t see anything. They talk about closed circuit television, except you couldn’t see it, because the picture was so unclear.
Donald Trump: (02:18:21)
You didn’t even know what they were doing. They could have been playing a baseball game. It’s a very sad thing for our country to have this, and they have to turn over the results. It would be easy for me to say, “Oh, let’s worry about four years from now.” No, this election was lost by the Democrats. They cheated. It was a fraudulent election. They flooded the market, they [crosstalk 02:18:48], and I just want to thank everybody for being there. This is a very important moment in the history of our country, and you’re doing a tremendous service to our country. Don’t worry about bravery, because the people that talk the most, they’re not the ones you have to worry about. These are all talkers, they intimidate, but these are not people that you’re going to ultimately have to … They push you around. They pushed our poll workers out. Our poll watchers were pushed out of the building. Okay. Some got back in, they were put in the pens, but these are not people. Don’t be intimidated by these people, but they’re bad people. They’re horrible people. They’re people that don’t love our country.
Donald Trump: (02:19:32)
We don’t have to worry about four years right now. We have to worry what happened on November 3rd, and previous to November 3rd. By the way, after November 3rd, when people put votes in and they put them in illegally, they put them in after the polls closed. One of our great Supreme Court Justices made mention of that. I can’t imagine that any Justice or anybody looking at it could be thrilled when they vote after the election is over. I want to thank everybody very much for being there. I want to thank the State Senate, respected people, tremendous people, and you’re doing a tremendous service for our country.
Donald Trump: (02:20:12)
If something was done wrong, if this election was won fraudulently and that’s what happened, it was a fraud. We’re talking about, very importantly, many more ballots, many more votes than the number we need. In other words, if we needed 50,000 votes, we’re not talking about, we found nine dead people that voted. Of course, there were many more than that, numbers that nobody even believes. No, we’re talking about numbers that are far in excess of the 50,000, far in excess of another state, where we lost by 10,000. They went absolutely wild, because we got far more votes than they thought possible.
Donald Trump: (02:20:52)
They’ve just stepped on the gas, and they got caught. Just like they got caught spying on my campaign, they got caught, exactly, they got caught doing this. I really appreciate it, and the country appreciates it. We have to turn the election over, because there’s no doubt. We have all the evidence, we have all the affidavits, we have everything. All we need is to have some judge listen to it properly, without having a political opinion or having another kind of a problem, because we have everything. By the way, the evidence is pouring in now, as we speak. I want to thank Rudy Giuliani for having the courage to do this, because there are other lawyers that backed out because they were being screamed at. Rudy is the greatest mayor in the history of New York, and there’s a reason.
Attendees: (02:21:40)
[crosstalk 00:13:41].
Donald Trump: (02:21:40)
He’s got great courage and he doesn’t care. He wants to do what’s right. I told him the other day, “Rudy, you were the greatest mayor in the history of New York, and you see what happened to New York without Rudy. You were the greatest mayor, but this is more important, what you are doing now is far more important than being a great mayor of the City of New York and being its greatest mayor by far. By the way, by far. This is going to be your crowning achievement, because you’re saving our country. Thank you all very much. Thank you.
Attendees: (02:22:08)
[crosstalk 00:14:13]. Trump, Trump, Trump, Trump. Woo! Trump, Trump, Trump, Trump, Trump, Trump, Trump, Trump, Trump, Trump, Trump [crosstalk 02:22:20].
Mr. Chairman: (02:22:09)
I think what you’ve just heard guarantees that a hundred years from now, that this is the most important public hearing ever held by this Senate committee. We turn now for our questions. Representative Ryan.
Senator Ryan: (02:22:45)
First of all, thank you very much, Mayor, for being here and for all the testifiers. Sixteen years ago this week, I was called out of retirement and recalled to active duty to go to Iraq, to do the elections in Iraq, in January 30th, 2005. That was an extraordinarily secure election. As a former commanding officer of a civil affairs group and psychological operations command, I know how to do elections. What happened in Pennsylvania, from a process control perspective, was absolutely a travesty. What I am concerned about is the remedy. How do we go from here? We see that there’s a perspective from our Council that in 1937, law provided the ability to appoint the electors to the secretary of state. I’m curious as to whether or not, Sir, you believe a special prosecutor should be appointed at the federal level to look at all of these irregularities, or whether or not there’s still actions that can be done at the state level to ensure that the goal of what we’re attempting to do, is to ensure, as we did for the people in Iraq and as I did for the people in Haiti, to ensure that the results are fair, accurate, transparent, and a representation of the will of the people of the United States.
Rudy Giuliani: (02:24:17)
Sound [inaudible 00:16:18]. My suggestion would be, and of course there are several different remedies that you could have, one would be to declare that the votes that were not inspected properly, be declared null and void in that county and deducted from the vote total. That’s essentially what they do in … Wisconsin has the strictest absentee law in the country. They require an application for every ballot. When they finish, they only count the number of absentee ballots that have an application.
Rudy Giuliani: (02:24:55)
They deduct from the total, from that County, whatever are missing. If you have 5,000 absentee ballots, but only 4,000 applications, you deduct 1000 votes. The reason they do that is to deter people from doing that in the future. Look at it this way, if you didn’t deduct the 678,000 or whatever it is, votes, that were not counted, there’s no penalty for it, what stops them from doing it again next time?
Attendees: (02:25:30)
Right.
Rudy Giuliani: (02:25:32)
This is a question of not voters acting improperly. It’s a question of the inferior offices of this state, because they’re inferior to the legislature in this regard, acted improperly. They conducted a completely sham election. They’re the ones who have to be penal … It’s a little like the exclusionary rule in criminal law. They’ve got to be taught, you can’t do that. The other thing you could do is you could demand to see all of the envelopes that were cast aside.
Rudy Giuliani: (02:26:06)
Like, I will virtually guarantee you’re going to see a couple of hundred thousand envelopes that have no names, partial names, the same name. You could ask to see the ballots. You’re probably going to see ballots that are official and ballots that are unofficial. You have two major gaps. You have the 672,770 votes that were not inspected by anyone secretly put into the ballot box.
Rudy Giuliani: (02:26:39)
Then you have this gap that I don’t understand, between the mail-in ballots you sent out, 1.8 million, and the number you counted, 2.5 million. That’s totally impossible to explain, other than what some of the witnesses were suggesting, that they were basically stuffing the ballot box. I believe what happened is they never expected to be behind by seven or 800,000 votes on election night. They expected to be behind by a couple of hundred thousand. In Philadelphia, to steal a couple of hundred thousand votes, they do it every year. That’s not going to be tough.
Rudy Giuliani: (02:27:17)
Now you had a real big problem, so you had to create mail-in ballots. You had to stretch it out for awhile. We didn’t go into detail on the Dominion system, but we have two other experts who can show you how they injected votes into the Dominion system. Also, so I think I would settle this election, but I would definitely appoint a special prosecutor to prosecute the people who conducted this election. You might even ask just a general question, how could the Governor and the Elections Commissioner, how could they have selected a company that counts our votes outside the United States?
Attendees: (02:27:56)
Amen. [crosstalk 00:02:27:58].
Rudy Giuliani: (02:27:57)
I didn’t know that.
Mr. Chairman: (02:28:00)
Thank you.
Rudy Giuliani: (02:28:01)
[crosstalk 02:28:01].
Mr. Chairman: (02:28:04)
Thank you. Representative Moul, questions for any of our panelists?
Speaker Panel: (02:28:07)
This is like our lightning round here to conclude.
Representative Moul: (02:28:12)
Thank you, Mr. Chairman. I think what I’m going to do is make more of a statement to throw out a timeline, because to try and reiterate what everyone else has already done, just be … A couple of years ago, our Supreme Court in Pennsylvania, very illegally redrew congressional maps. It’s very clear in our Constitution, that is a legislative act. Fast forward, couple of months ago, our Supreme Court rewrote our election law. Again, that needs to be done in statute. It is a legislative act.
Representative Moul: (02:28:55)
When it came to the mail-in ballotings, that House Bill, I want to say it was 77, Act 77, I was really kind of taken aback by how eager the Governor was to sign that Bill. Normally, Bills can sit in his desk, eight, and almost 10 days before he signs them or lets them become law on their own or vetoes them right away, and that one he signed almost instantaneously. I just kind of wondered. After I saw what happened in the primary, I wrote House Bill 2626, and this is where it kind of locks it in for me, Mr. Mayor and panelists. In House Bill 2626, I saw where there could possibly be stuffing the boxes, so I said simply, “Let’s put a barcode on the bottom of every mail-out ballot, so that the machine has to read a barcode that was accepted by the elections office.”
Representative Moul: (02:30:00)
Before that ballot goes out, they zap it. When it comes back, if that barcode doesn’t match one of the barcodes that they sent out, it gets kicked out. The Governor immediately, as soon as we ran that Bill out at Committee, said, “That hits my desk, it’s an automatic veto.”
Attendees: (02:30:18)
Wow. [crosstalk 02:30:19].
Representative Moul: (02:30:19)
Not everybody knows that, but that came to us. We had to literally strip that out of the Bill. I will be re-introducing a Bill to barcode any ballot that gets mailed out, when we get back. Trust me. Then we have the drop boxes, the three days extra to vote, ignoring the signatures and ignoring the postmarks, and I have to come to the conclusion that you did also. If it looks like corruption, smells like corruption, it’s corruption, and we’ve got to get to the bottom of it. Thank you for being here. Thank you for your testimony. Appreciate it very much. Thank you, Mr. Chairman.
Mr. Chairman: (02:30:59)
Thank you. Senator Regan.
Senator Regan: (02:31:02)
Thank you, Mr. Mayor. Thank you so much for being here. Thank you to all our testifiers today. It’s been very informative. My question, Mr. Mayor, you touched upon it briefly, but I want to just, if you could delve it a little bit more into it, Attorney General Barr, shortly after the election, authorized the US Attorneys across the country to investigate allegations of voter fraud. I wonder if you could tell us about the criminal investigations that are going on, maybe specifically in Pennsylvania, by our US Attorneys and maybe our FBI, and tell us how that is going? Is there any evidence that’s been presented to you or are you made privy to any of this information?
Rudy Giuliani: (02:31:39)
I’m sorry, Senator, I wouldn’t be made privy to the criminal investigations. In my experience, criminal investigations would take too long to help you in your most immediate decision. Criminal investigations could help you in your longterm decision as to how to reform, so this never happens again, which I think you all will want to do. For example, not ever hiring again, a company that counts votes in Frankfurt, Germany, that’s owned by two Venezuelans, who are very close to Hugo Chavez and Maduro. That’s extraordinary.
Rudy Giuliani: (02:32:19)
Unfortunately, we don’t have the time to really analyze the votes. Although, if you want to go into greater detail, we have other forensic experts that can show you votes that were specifically put into the machine, because these machines have a back door that allows you to do that. I would say the criminal proceedings are going to take six months to a year. What you have before you, that you can verify, is you have before you, very, very clear evidence. It’s not small. It’s not as if these are a couple of little mistakes that happen in … I hear people say, “Oh, every election has a couple of little mistakes.” Not 682,000 ballots, that nobody got to inspect, that from time immemorial, we’ve never had trouble in America with absentee ballots.
Rudy Giuliani: (02:33:09)
I’ve been in absentee ballot contests in New York, and they can be pretty vicious. The language is worse than that poor lady experienced, but we would never think of having one without the Republican there and the Democrat there. Even in New York, we would never think of it. Here, they did it 700, 000 times. That’s an insult to the legislature, because you didn’t provide for that, and you’ve got this gap with 700,000 unexplained mail-in ballots, that come out of nowhere. I think you have more than enough to say that this election, the numbers don’t add up.
Rudy Giuliani: (02:33:52)
It’s easy to figure out what the right numbers are, by excluding the illegal votes and count the honest votes, and the winner of the election changes.
Senator Regan: (02:34:02)
Thank you very much, sir.
Rudy Giuliani: (02:34:03)
Thank you.
Mr. Chairman: (02:34:04)
Senator Mario Scavello.
Senator Mario Scavello: (02:34:10)
I’m just going to make a couple of comments. First, I want to thank everybody for coming out here today. We need your support, your continued support throughout this whole process, because we’re going to also get the other side that don’t want us to do anything at all, and we definitely need to do something. We need to correct this problem. My Northampton County Chairperson, Lisa [inaudible 00:02:34:34], did an excellent job explaining what was going on in Northampton County.
Senator Mario Scavello: (02:34:38)
One of the biggest things that bothered me more than anything else, was people that went back, went to vote and they were told they already voted. That happened, I must have heard it from 50 people in my district. They made them sign a provisional, and I called up and I says, ” You better make sure that those provisionals are counted.” Some of the other things that were happening, most of the stuff that you said out there, it happened in every county. When you didn’t want to vote with your absentee and you went in, you’re supposed to get a ballot, not a provisional.
Senator Mario Scavello: (02:35:22)
There were people out there that got provisionals, but just a little information for all of you, the new machines, this new process, we didn’t vote for it. That was something that was pushed by the Governor. He mandated that all of the counties had to go and change their system, because he wanted some type of what we don’t have, is a form, to see who voted. They want a certificate or something, a slip to see how you voted, who voted. It’s obvious that this system has not worked, and the people of Pennsylvania are not happy about it. Thank you.
Mr. Chairman: (02:36:11)
Thank you, thank you. Madam Majority Leader Elect?
Senator Kim Ward – Madam Majority Leader Elect: (02:36:19)
Thank you. First of all, I just want to say what they did in Philadelphia, when they closed us out and made us go to court to do what we should have legally been able to do, is a travesty to what we in Pennsylvania, need, to have confidence in our elections. That should never happen again, and we should probably tighten our laws so that it can’t. Secondly, I’m extremely bothered by the absentee ballot and the mail-in ballot request number being almost 700,000 off, from what we received.
Senator Kim Ward – Madam Majority Leader Elect: (02:36:53)
We just did try to get on the Department of State, and they did take down their dashboard, so we can’t even check it. If you have any of that information, any pictures or any website downloads, we would really appreciate having those, because they took them down. I don’t know. Everything they do, it looks like they’re trying to hide something, and really, really continues to erode any kind of confidence in our process.
Rudy Giuliani: (02:37:17)
Senator, what I would suggest is, they decided that morning or maybe a couple of days before, to exclude you, because they knew what they were going to do. It wasn’t an accidental sort of whim to exclude all Republicans. It’s unheard of to exclude all Republicans. They excluded you because of what you would see. You would see ballots that weren’t properly filled out. You would see ballots that were filled out at the last minute. You would see them start to get reports of how far behind they were, because they were connecting to Dominion and Smartmatic, and they were telling them how far behind they were.
Rudy Giuliani: (02:37:57)
You would have caught them right in the middle of the fraud. Why create this big problem for yourself of excluding Republicans in six states, hundreds and maybe thousands of them, if you’re not doing something wrong? If you’re doing something as the Senator said, that’s kosher, come on, look, who cares? This big thing wasn’t done because they were conducting an honest vote. It’s kind of crazy to think that. This was done because they knew this is the only way they could steal this election. When he was ahead by those numbers on Tuesday night, Wednesday morning, they panicked.
Rudy Giuliani: (02:38:34)
I’ll also tell you there’s an expert witness who can testify that this pattern fits the pattern that Smartmatic used in two elections that they stole from South America. When the election was getting too far away, they called a halt in the voting count. Well, there was a halt called in six places, including in Pennsylvania. In one place, they made believe there was an overflowed water tank. It happened to be one little toilet bowl. They took a two hour halt, and in Michigan, they brought in a hundred thousand ballots through the back door and counted them with nobody observing them, except three people who happened to be remaining behind.
Rudy Giuliani: (02:39:18)
That’s evidence that’s also in affidavits and could be presented to the Michigan Legislature. Those people are willing to testify. Just one other thing, these people are very brave people. A lot of people are testifying. They’re backed up by 50 more who are willing to testify. Obviously, we can’t present all of them. I want you to know that all of them are backed up by numerous witnesses who say the same thing, but there are probably another 50 who are not willing to testify, including probably what would have been one of our best witnesses, because he’s been threatened with the loss of his job and his children being thrown out of their private school.
Senator Kim Ward – Madam Majority Leader Elect: (02:40:00)
Thank you very much, Mr. Mayor. Thank you.
Kim Ward: (02:40:03)
Thank you very much, Mr. Mayor. Thank you.
Senator Argall: (02:40:04)
Thank you, Senator. Let’s try our Zoom capabilities here. Senator Michele Brooks, calling in from Northwestern Pennsylvania.
Michele Brooks: (02:40:15)
Thank you, Senator Argall. I did want to make two comments first. I join my colleagues in being concerned about the arbitrary Supreme Court decisions and their decision to decide to be legislators. And all Pennsylvanians need to be concerned about this. It’s not only the election or redrawing maps, what’s going to be next that impacts our state? And so not only do we need to be looking at what happened with this election, we need to be looking at how we can stop the Pennsylvania Supreme Court from wanting to legislate when that’s not what they’re supposed to be doing. And it’s very disconcerting the decisions they made contrary to state law in regards to this election.
Michele Brooks: (02:41:15)
My question is, if you could talk about the drop-off boxes, again something the Pennsylvania Supreme Court allowed. Talk about the security around them. I know in my area, I did not have a significant amount of drop boxes, but can you talk about what type of security or parameters of safety were around these drop-off boxes and also how many ballots were collected in these drop boxes? Thank you.
Rudy Giuliani: (02:41:52)
That of course is the subject of another lawsuit that was brought the other day. I only know it generally, the drop-off boxes were provided, speaking very broadly, in the Democrat part of the state, not in the Republican parts of the state. There were many, many, many very close to each other in Philadelphia, in Allegheny County, places like that, they were miles and miles apart in other places. And as far as I can tell, I’ve only read the lawsuit, I don’t know the details of it, there was no security for them. And there are many situations where very suspicious activity was seen with people dropping off, you’re not supposed to harvest votes, people were dropping off 30 and 40 ballots at a time. That’s a pretty big family.
Rudy Giuliani: (02:42:48)
They’re going to have a tough time this Thanksgiving, by the way, with [inaudible 00:02:51]. They’re going to have to eat in like four or five different tents with, hopefully there’ll be able to celebrate Thanksgiving, but I’m sorry. I don’t know the details of that, but it is the subject of another lawsuit and the decision of your Supreme Court that is completely outrageous is the one in which they say that present means just being there. I mean, there was a movie with Peter Sellers called Being There. I don’t know if you have a saw it but he was a potted plant. That’s totally absurd to think that when you passed the rule that there should be present representatives of both sides or all sides that you meant for them to sit there and look at the drapes. I mean, they were supposed to be there as the intermediate court decided.
Rudy Giuliani: (02:43:40)
This case was decided by the intermediate appellate court in the president’s favor with a very sensible decision saying present in this context can mean only one thing, present in order to see the identifying data. And in a completely political decision [crosstalk 02:43:57], they ruled what I would regard as it was totally irrational ruling, which we will appeal to the Supreme Court. I think it was embarrassing-
Senator Argall: (02:44:08)
Thank you.
Rudy Giuliani: (02:44:09)
… for them to rule that way.
Senator Argall: (02:44:12)
Thank you.
Senator Argall: (02:44:12)
Senator Judy Ward.
Judy Ward: (02:44:19)
Thank you. I also want to thank our testifiers today. They were incredibly brave. And what I heard today was so troubling to me. I don’t know if I’ll ever get it out of my head, and I’m sickened by the fact that it happened here on our watch. In regards to doing a forensic audit, would this be able to be done or be anywhere accurate? Does anybody know?
Rudy Giuliani: (02:44:53)
I was just looking around for my two experts. He came over to tell me that all of the material that we have, forensic material we have is archived and we can easily make it available to you online. And you can look it over. I don’t understand all of it, but maybe I have people help me understand it, but yes, you could do a forensic audit of the machines.
Female: (02:45:17)
Do it.
Rudy Giuliani: (02:45:18)
You could do that.
Senator Argall: (02:45:20)
Thank you.
Rudy Giuliani: (02:45:24)
I think it would be very, very helpful. I understand that even one machine would give you a great deal of information about how the whole system operates and how they slip votes in at various times. And you’re not supposed to be connected to the internet in the counting room, but they were connected to the internet. And your tally was being sent to Frankfurt, Germany, where it was being analyzed.
Senator Argall: (02:45:51)
We have a question from Senator Langerholc out in Johnstown connected via Zoom. Wayne, can you hear me?
Wayne Langerholc: (02:46:00)
Yes.
Senator Argall: (02:46:00)
Thank you.
Wayne Langerholc: (02:46:01)
Can you hear me?
Senator Argall: (02:46:01)
Yes loud and clear.
Wayne Langerholc: (02:46:02)
Thank you. Thank you, Senator Argall. Thank you, Senator Mastriano. I thank the testifiers for being here. Mr. Mayor, thank you for your testimony. I’ve been vocal that we must allow this legal process to play out and a country that we live in that we do not allow that is not a country that I want to be a part of. My question is on that legal review and on those legal process.
Wayne Langerholc: (02:46:25)
Mr. Mayor, can you give us an indication of how many lawsuits are pending, where they’re at and the various appellate processes? And second part to that, the allegations of the mail-in ballots, the 1.8 versus the 2.5 that came in, is that the substance of a lawsuit? Thank you.
Jenna Ellis: (02:46:47)
This is Jenna Ellis and the Mayor has asked me to answer this question. So as far as the campaign is concerned, we have pending lawsuits here in Pennsylvania that’s going up to the third circuit that granted an expedited review. We also have pending litigation in Nevada. We also have recounts in Georgia and Wisconsin, and we also have a pending lawsuit in Michigan. So regarding the mail-in specifically, we are still looking at all of the other legal options, and we are also planning on filing additional litigation by the end of this week. I won’t go into detail on that other than to say that this is asking for a judicial remedy to this.
Jenna Ellis: (02:47:31)
Also, I’ll invite the question from the panel as well, in terms of a remedy. Part of what we are asking, or what we did ask initially was to stop certification so that we could get to the bottom of this process. Clearly you’ve heard sufficient evidence today that would lead any reasonable finder of fact to say that there’s been sufficient evidence that you should stop that, that you should not move forward with certification. What we would ask the judiciary in terms of remedy can be legislative as well under the constitution. And I would direct your attention to Article 2 section 1.2, which says that each state shall appoint in such manner as the legislature thereof may direct a number of electors.
Jenna Ellis: (02:48:13)
And so you, the legislature without judicial oversight can direct and take back that power. You have the opportunity as the legislature and legislature in this context means the state legislature. So although there has been delegated authority to your secretary of state and in according to your law at any time, you can take back that power. And that is the provision and the protection that is embedded in the constitution to make sure that when there are instances of corruption in a state that the state legislature, which is the representatives of we the people, all of these wonderful people who are here in Pennsylvania and all of your constituencies who voted and who want their voice to be heard. Because we are a government of the people, by the people, for the people, we the people need to submit, need to submit our vote.
Jenna Ellis: (02:49:11)
And so I would say in terms of a remedy, you may fashion that even outside the purview of the litigation that we are now bringing, and I’m happy to go into that into more detail if the panel would prefer.
Senator Argall: (02:49:24)
Thank you, Representative Kauffman.
Kauffman: (02:49:28)
Thank you very much. And thank you, Mr. Mayor for being here and Attorney Ellis. We appreciate your service to the nation because these are definitely difficult times. And all of those who have testified, it truly does take bravery to step out in situations like this and talk about your personal experience.
Kauffman: (02:49:51)
I wanted to direct this to Mayor Giuliani and Attorney Ellis. In the cured ballot issue, which or uncured ballot issue, the ability to cure in one area of the state and not in others, there is certainly to many of us seems to be unequal protection violation under the 14th Amendment. Can you quantify what you’ve found as far as maybe number or information as far as the number of ballots we’re talking and how widespread it is across the commonwealth, as far as, were there two counties that allowed for curing and, they were Philadelphia and Pittsburgh, or were there more counties out there that allowed for curing? That kind of information would be very helpful to us, I think.
Rudy Giuliani: (02:50:51)
There’s no question representatives that it went on. It went on in the two counties you mentioned and probably three or four more. Curing, in other words, it did not go on in the majority of counties, Republican or Democrat. Most of them were in [inaudible 02:51:09]. The elections commissioner sent out a notice advised that you could cure the ballot. If you check with your lawyer, your lawyer would say, “No, no, you can’t do that. The legislature has to provide for a cured ballot.” So your counties that were sticking to the strict letter of the law didn’t provide for curing. The counties that were acting, I would say illegally because the election commissioner doesn’t have the authority to make that change followed it. It does turn out that those are largely the Biden counties did the curing and the Trump counties didn’t do the curing.
Rudy Giuliani: (02:51:51)
It’s hard to know how many it affected. I could get you the number of actually cured ballots but I don’t think I can get you to the number that weren’t cured, who are the ones that were effective. And when argued this in front of the court, the judge said, “Well, maybe those people in the counties that didn’t allow curing should sue that county,” but that County didn’t do anything wrong. That county followed the law. The reason they were deprived of their right to vote on unequal basis is because other counties followed what would be the illegal advice of the Election Commission that you could cure a ballot for which there is no authority in state law.
Rudy Giuliani: (02:52:38)
So we will try to do better to get a good number there, but it’s hard to do because a lot of it affected people who just didn’t vote.
Kauffman: (02:52:48)
Sure. And-
Rudy Giuliani: (02:52:49)
But I do think the practice, in some cases the numbers are sufficient to overturn the election. In some cases, the numbers aren’t but what they do do is show how onerous and how draconian the measure they took to flip this election. I mean, some of them result in numbers that affect the outcome of the election like not examining the ballots, some of them don’t, but they’re still indications this was a deliberate fraud. This wasn’t just a couple of election officials kind of screwed up. This was a deliberate planned fraud. And that’s why the remedy, which seems kind of tough is justified.
Rudy Giuliani: (02:53:35)
If this were just a couple of mistakes, that would be a tough remedy, but this is a plan that was carried out throughout your entire state. And I submit it was carried out in concert with other Democrat organizations. And if you look carefully, they picked only Democrat cities, where they control law enforcement and the courts, and they can get ridiculous decisions like the first the decision they got from a judge in Philadelphia, who said, “Oh, you are present. Why do you have to see anything?” Well, because I’m an inspector. That’s why. That’s a purely political hack decision. And they knew they could get decisions like that in Philadelphia, in Detroit, if it were a different kind of election, Chicago, New York. Those are the kinds of cities they picked. They didn’t pick even in your state kind of neutral cities, Democrat, or Republican.
Kauffman: (02:54:34)
Thank you.
Senator Argall: (02:54:34)
Thank you. Representative Schemel.
Paul Schemel: (02:54:38)
Thank you, Mr. Chair. Three quick questions for three different testifiers. So we certainly know about the irregularities and inconsistencies you just detailed and inconsistency and you can’t prove a negative that’s why you can never prove votes that were never allowed to be cured. But in regard to allegations of fraud, I have questioned first and forgive me, I don’t remember names, but the gentleman here from Philadelphia who was denied admittance to observe in a poll.
Paul Schemel: (02:55:02)
You described being denied the ability to get into a polling place. Did you seek to be admitted to any other polling places and were also denied?
Speaker 10: (02:55:10)
I could have gone to other polling places where I heard there was problems, but we decided to move on to other problems. What was her name? I was actually at her polling place when she was being totally harassed, I was one of about five, six guys to stop the harassment-
Paul Schemel: (02:55:31)
So there was no polling place that you sought admission to in and around the city of Philadelphia that just allowed you in, when you displayed your orange card?
Speaker 10: (02:55:38)
I probably could have gone to that entire ward and had the same problem. We went out of that ward because there was other problems going on that day.
Paul Schemel: (02:55:48)
Okay. Next question for a gentleman next to you from the attorney from Florida, you were observing polls and the processes in the city of Pittsburgh. I believe that you described being kept away from being able to observe the tabulators and so forth. That was a problem we certainly saw in other places, in the city of Philadelphia. When you were eventually allowed to get closer where perhaps you could see the ballots and the votes, did you observe anything different about the behavior of the tabulators? Did their behavior change? Did their methodologies change? Anything to lead you to think that they were conducting the process one way when you weren’t observing and a different way when you were?
Speaker 11: (02:56:26)
In the first instance, we were never really able to get… There was only one element of the process where we were able to actually get reasonably close to see what a tabulator was doing. And that was with the overseas and military ballots. When overseas and military ballots come in, they come in a different format that’s not readable by the machines. And so there’s a individual that takes the ballots and then keys in the votes for the voters. And of that group, we could see about three or four of those people doing that transposition of those ballots.
Speaker 11: (02:57:07)
Beyond that… And that was for a very short period of time, and actually Friday evening, Friday after the election, they were beginning to tabulate overseas and military ballots. With that singular exception, we couldn’t actually see any ballot at that particular moment that you ask about. There was no opportunity to ever really see any ballot other than that situation. And there was one worker that was within 8 or 10 feet of us at the edge. And we did in fact see him do 35 Biden military ballots on a row, which we found to be kind of curious, but what he was doing did equate with the papers that he had in front of him.
Paul Schemel: (02:57:53)
Okay. Thanks. My final question for Mr. Giuliani. You’ve alleged fraud within our voting system and we’ve heard a lot of anecdotal evidence we have as representatives and senators, and you’ve presented a good deal today. But amongst the affidavits you have, do you have any affidavits from anyone who could be quantified as a whistleblower who actually was part of the fraud and is now willing to testify as to the fraud?
Rudy Giuliani: (02:58:18)
Yes, we do. We have, I believe… I have to check this, but I believe sometimes I get the states a little confused, but I believe there are three that would need a subpoena to protect themselves in order to testify.
Paul Schemel: (02:58:39)
Okay. And then I would presume that’s part of the legal-
Rudy Giuliani: (02:58:40)
I would say at least one of them is a Democrat. I’m not sure the other two.
Paul Schemel: (02:58:45)
… that’s part of your legal claims I would assume.
Rudy Giuliani: (02:58:48)
Yes, sir.
Paul Schemel: (02:58:49)
Very good. Thank you.
Rudy Giuliani: (02:58:49)
And if there’s a subpoena process, we could work with them to come forward. They’re frightened for their jobs and I don’t want to make too much of it, but they’re also frightened for their physical safety. I mean, this is a case in which one of the lawyers, one of our chief lawyers in the case had to leave because his family and his little children were threatened for representing the president of the United States. That’s not America.
Senator Argall: (02:59:16)
Representative Jones.
Mike Jones: (02:59:23)
Can you hear me? Okay, great. Thank you, Mr. Chairman. I appreciate it. Thanks to everyone who testified. I do have a question, but a couple just comments to sort of get on the record. For those that maybe think this is like, we just don’t like the result, that’s not really the case. I’ve spent weeks since the election having numerous people go through data. And for the sake of time, I just want to highlight a couple of examples why some of these things just at a minimum, they certainly make you scratch your head a little bit.
Mike Jones: (02:59:57)
One thing is voter registration trends that we went back to 2004 and all of the presidential elections, and pretty much without exception, the registration trends in a given county are a pretty good indicator of how the votes would go. Statewide for example, Democrats had a 1.1 million, I’m rounding the numbers here in 2012, about a 1.1 million voter registration advantage. And then President Obama won re-election. In 2016, that number was down to 900,000, just under 900,000. And as we know, President Trump won Pennsylvania by about 44,000 votes.
Mike Jones: (03:00:34)
In the weeks leading and the months leading up to the election, Republicans were gaining about five to 10,000 votes registrations at least per week. I know that because I was spearheading our York, Pennsylvania for Trump effort in my spare time. And what had been a 900,000 advantage at election day was down to about 680,000.
Male: (03:00:57)
Wow.
Mike Jones: (03:00:57)
That’s a couple hundred thousand almost all of which occurred in the six months or so leading up to the election yet the president loses by 90,000 votes. Not impossible, just want to get it out there. Let’s drill down a little further. My home county of York County, which netted more votes for the president than any other in 2016, we had a 13,000 increase in Republican registrations. Democrats dropped a thousand, Republicans increased 12,000 yet the president’s margin of victory decreased by 2,000.
Mike Jones: (03:01:32)
In Westmoreland County, there was a 26,000 votes swing, a county even smaller than York. Democrats saw a 10% decrease in registrations yet Joe Biden outperformed Hillary Clinton by more than 20%. It doesn’t prove anything, but I want to at least bring it to everyone’s attention that we’re just not sort of making things up here that we don’t like. Some of these are certainly head scratchers. When you lose 10,000 Democrat registrations gained 16,000, yet the margin of victory stays the same and Biden increases by 12,000 votes in that County, that’s surprising.
Mike Jones: (03:02:10)
Lastly, if I may, I want to talk a little bit about… I don’t know if the gentleman can testify or can return, but your witness on… The gentlemen with the military background on the voter fraud and so forth.
Male: (03:02:23)
[inaudible 00:22:24].
Mike Jones: (03:02:24)
Yes. Thank you, sir. I guess my question here is, do we believe, or have reason to believe that the mail-in would be more or less susceptible than the election day vote to manipulation? And here’s why I ask another head-scratcher. I’m comparing Republican to Republican, Democrat to Democrat. We all know that Biden did better with mail-in, and that was expected because a lot more Democrats voted by mail. And of course the president did better on election day. Nothing to see there. However, when we compare President Trump to a relatively unknown candidate named Stacy Garrity, who was elected, a Republican who won the race for treasurer, on election day, the president did 128,000 votes better than Ms. Garrity on election day. Not really all that surprising number of people don’t vote down-ballot and so forth yet with mail-in ballots, she did 41,000 better than the president.
Mike Jones: (03:03:28)
I have a hard time understanding that. Let’s go to Joe Biden. We have a relatively well-known attorney general here, Democrat, Josh Shapiro, who won reelection. He on election day, he defeated Joe Biden, 52,000 votes. He did 52,000 better than Joe Biden yet with the mail-ins Biden does 54,000 better than Shapiro. I just sort of want to get that on everybody’s radar. Maybe there’s an explanation, but I don’t really understand how Democratic mail-in voters are that much different than Democratic election day voters. And interestingly enough, apparently Republicans who vote by mail are much more likely to vote for the down-ballot candidate than on election day.
Mike Jones: (03:04:19)
Last comment there, part of the argument is that people didn’t vote down-ballot. That’s going to be a lot more prevalent on election day when you’ve waited in line for two hours, and you’re in a hurry to get out there. But this speaks to the opposite. With a mail-in ballot, you have plenty of time to complete your ballot and to go down-ballot. So if that was the case, the opposite should be true if you’re tracking with me. So I guess my question there is on, do we think the mail-in is more susceptible?
Mike Jones: (03:04:50)
And then lastly, maybe for you, Mr. Chairman, or whoever has a little more clout here than I do, we need, it is imperative that we secure the sure system data. And some of the things that we are questioning can be easily proven or disproven. We just want the facts. So for example, if we see huge numbers of inactive voters, I think one of the witnesses spoke to that, did we see huge numbers of inactive voters on the mail-in side? That’s all right there in the data. We know how frequently people voted. We know who requested mail-in ballots. Did we see multiple mail-in ballots going to the same address?
Mike Jones: (03:05:34)
It is mind boggling the record turnout that we saw in a state where mail-in voting is brand new during the time of COVID where the ground game was certainly hindered by that. Doesn’t mean it didn’t happen, but we have the data. This is a simple query of data. This is not a manual recount of ballots. This is a simple query of data that the state possesses specifically to say, did significant numbers of mail-in ballots go to the same address? I could do that myself in probably 10 minutes if I had the data. And did we see large numbers of inactive voters coming out for a candidate who had very little enthusiasm? And maybe it all happened, but we don’t have to speculate, the data’s there. And I would suggest we have a look at that.
Mike Jones: (03:06:27)
Thank you for humoring me with that. It’s just a topic we hadn’t touched on very much. I wanted to throw some examples out, and I am curious if you feel the mail-in is more susceptible or less susceptible than the in person voting?
Phil Waldron: (03:06:38)
Representatives Jones, the user’s manual for specifically the Dominion and the ES&S suites, they do allow the administrator to batch process mail-in votes. So what that does is that they process all the mail-in votes in a batch, and it can be a batch, they normally scan in batches of 50, but they can just chunk all of those votes into a batch file. The administrator then can select which candidate or what percentages of that mail-in ballot go to which candidate. So they’re very susceptible to individual manipulation by the administrator operator.
Mike Jones: (03:07:17)
If you were trying to move large numbers of votes, would it be more logical to do that through the mail-in where you had a large batch at a county level or am I speculating too much there?
Phil Waldron: (03:07:29)
Yes. To do a manual recount of all those ballots is much more time-consuming. And like we mentioned earlier, if they were mass produced, and they were single vote, then there is potential to add extra ballots to the county.
Mike Jones: (03:07:46)
Thank you very much and I appreciate you giving me the time Mr. Chairman. Also, I’m just curious what we heard the testimony with the USB sticks in Delaware, why that’s not being investigated. That seems like that would be criminal activity that we’ve heard here discussed and whether where law enforcement is on that. Probably not your immediate concern Mr. Mayor, but I’m curious about that as well. Thank you very much.
Senator Argall: (03:08:13)
Thank you. Representative Zimmerman.
Dave Zimmerman: (03:08:15)
Yeah. Thank you, Mr. Chairman, and thank you Mayor for coming to Pennsylvania and all those willing to testify, thank you so much. So just to comment, and then also a question. So in Lancaster County where I’m from, since May, we registered over 12,000 new Republicans and a large percent of those ended up being Amish. They might understand the difference between freedom and socialism more than most of us, but I think probably the audience is here for that reason as well.
Dave Zimmerman: (03:08:51)
That’s just a comment just for your information. The question I have, Mayor for you is that as legislators here in Pennsylvania, is there a one or two things that you would advise us or suggest to us that we really concentrate on or make to make a change? What comes to mind that would be most important for us to really do here in Pennsylvania?
Rudy Giuliani: (03:09:19)
First thing, of course, as a lawyer for my client, along with Jenna, we would ask you not to certify and to certify the correct honest votes, which I think can be arrived at several different ways. And probably the easiest way to arrive at it, just because of the huge number of votes, are the votes that were not properly inspect it. And when I say properly inspected, they weren’t inspected at all. You could almost consider them hidden votes, counted in a back room where everybody was smoking cigars.
Rudy Giuliani: (03:09:53)
And the reason you have a lot of votes without a down-ballot, you’re quite correct, Representative Jones, that it’s much easier to fill out your home. You can fill out the down-ballot than rushing through a machine. However, if you only have a half hour to get 10,000 votes in, you don’t have time to do the down-ballot, do you? If you’re sitting in a truck somewhere right near the place you’re going to bring in shopping bags… And I don’t think we have this evidence here, but we have this evidence in Michigan. We have evidence of trucks coming in with ballots in garbage pails and ballots in cardboard boxes.
Rudy Giuliani: (03:10:34)
So maybe it’s my prosecutors paranoia, but it seems to me that those ballots were filled out that night. And you don’t have time to vote for all those. You just got enough time to go… And that’s why you get this strange anomaly that there is so many down-ballots not filled out for Biden.
Phil Waldron: (03:10:57)
Can I add one thing?
Rudy Giuliani: (03:10:59)
Yes, sir.
Phil Waldron: (03:11:03)
I learned as a lieutenant never to bring a problem to my superiors without a solution. That’s one of the things I try to live to today. There are solutions that are available. I made the simple comment that your vote should be at least as secure as your Venmo account and it’s not. It’s easily fixable with app, with blockchain. There is a process out there that was developed by MIT. It’s called Voatz, V-O- A-T-Z. It’s being fielded now. The problem is there are no national standards for federal elections, and there are no enforceable standards. So if nothing else out of this election, if we have a set of enforceable standards and we use a technology like blockchain to ensure that every vote going through the whole process is transparent from soup to nuts, then we can ensure a technologically advanced secure election that’s not… An individual maybe manipulable but-
Speaker 12: (03:12:02)
An individual vote may be manipulable, but batches of votes would not be, so.
Speaker 13: (03:12:06)
Great. Great information. Thank you so much. Thank you, Mr. Chairman.
Senator David Argall: (03:12:09)
Thank you.
Jenna Ellis: (03:12:10)
Can I add one thing as well for the mayor?
Speaker 13: (03:12:12)
Oh, sure.
Jenna Ellis: (03:12:13)
So you asked what two things. Well, I think those two things are very clear that you have to deal with today and then you have to deal with the future. And the more pressing instance right now is today, and this body should deal with that in some way. And you are the constitutionally vested entity to do that. You have a variety of options in front of you. And when you deliberate together how to best address that, you have the constitutional authority and permission to do it. And so the mayor has suggested one possible remedy. There are others. And you could call for a special election, still. You can direct the manner of your electors. You have a variety of constitutional options, but one option should not be to ignore it and to certify a corrupted, irredeemably compromised election.
Jenna Ellis: (03:13:12)
So we would ask that you deal with that today. And then for the future, look at all of these things that you’ve raised. Look at all of the voting machines. Look at all of these people who are interfering, really. You have an executive office and branch that is runaway. You have from your governor to your secretary of state, to each of these individual election officers. This is election official fraud, and that’s what’s going on here. And for the sake of every future election, that should be addressed in the legislature, because you also have the constitutionally vested obligation to the people of the state to address that.
Jenna Ellis: (03:13:52)
So my recommendation, along with the mayor, and I sincerely applaud everything that he has said as well. And on behalf of our client, but on behalf of the nation, you’re right when you say that this isn’t just about overturning an election. It’s not about deriving the outcome. That’s what the fraudsters want to do. What we want to make sure of is that we the people get to select and prefer our Commander in Chief, and you have the obligation and the responsibility to ensure for the people of Pennsylvania that that occurs.
Senator David Argall: (03:14:20)
Thank you. Thank you very much. I believe we have Representative Metcalfe on Zoom with one final question. Daryl.
Rep. Daryl Metcalfe: (03:14:31)
Can you hear me, Senator Argall?
Senator David Argall: (03:14:32)
Loud and clear.
Rep. Daryl Metcalfe: (03:14:35)
Thank you, Senator Argall, Senator Mastriano, for hosting this very important hearing. It’s very important to the future of our Republic today. Thank you for the invitation to the House members that are joining you there today. Thank you for the invitation for me to join you via Zoom today, and thank you for the opportunity to provide some input and ask a question of Mayor Giuliani here today. But a lot of what’s been talked about, and I really appreciate Mayor Giuliani and the attorneys [inaudible 03:15:09] to the General Assembly to [inaudible 03:15:12] our constitutional authority regarding the certification of this election.
Rep. Daryl Metcalfe: (03:15:19)
I am a former state government committee chair that had oversight for the election law. For many years, I served in that position, not during the class term, but in the past. And we have battled in Pennsylvania to fight the fraud and fight the way that our election laws have been violated, not just for several cycles, but for decades. And we’ve actually had a state Senate race that was overturned, I believe in the Philadelphia area, back in the nineties because of fraud. So when we look at what has occurred here and really I appreciate Mayor Giuliani bringing all of those individuals before us today to provide a testimony at this hearing. And I think even more important, I hope that we get a chance to see that their testimony was heard in a court of law where a just ruling is found to protect every legal vote that was cast here in this election in November, and to discard all those votes that were not legally cast.
Rep. Daryl Metcalfe: (03:16:25)
And I think as we’ve been overwhelmed, as was mentioned by the senators at the beginning of this meeting, that as we came through election day, and prior up to that, we’d had about three and a half million people filing for unemployment because of the unconstitutional dictates of Wolf to shut down businesses and stop people from going to work in Pennsylvania. And we were just overwhelmed with those calls. But since election day, we have been overwhelmed with calls from individuals who are distraught, distraught over the future of our Republic, the future of our future elections, and being able to ensure that people are having their legitimate legal votes counted to elect who’s going to govern them. And right now with the examples that we’re shown here today with these witnesses, a lot of the people that have been calling my office and emailing me, they are demanding action.
Rep. Daryl Metcalfe: (03:17:23)
And as we’ve heard today, there’s a lot of citizens out there that have important information that needs to be shared in the courtroom to ensure that these fraudulent activities, and I mean, a lot of times when people talk about fraud, they’re thinking of impersonation. But that’s not what we’re talking about. We’re talking about fraudulent activities as far as a fraudulent election, because they’ve not abided by the laws that have been passed by we the people through our elected legislature, as the U.S. Constitution, as our State Constitution, as our laws provide for to facilitate our elections.
Rep. Daryl Metcalfe: (03:18:05)
So Mayor Giuliani, as we move forward. And I stand with Senator Mastriano and others that are there today. I actually sent an email out while I was listening to some of the testimony today given at the hearing to the whole Republican caucus in the Pennsylvania House to encourage them to sign on and listen to what was happening today with this information that I think they all need to hear, and that every Pennsylvanian, every American needs to hear. To help encourage our citizens, Mayor Giuliani, as you move forward in the court battle, I know the press has been continually day after day saying no evidence, no evidence, no evidence. Well, I think we heard a lot today. Do you expect that you’re going to have an opportunity in the right level of courts in our state and our nation to have this evidence presented by some of these people here today and others that you have signing affidavits?
Rudy Giuliani: (03:19:02)
Yes, I am. I’m certain that there will be a court decision that will allow us to present our evidence. The case in Pennsylvania was dismissed on a motion to dismiss in which the judge made factual findings against us, basically saying he thought our claims were absurd. Of course, he has no right to do that on a motion to dismiss. On a motion to dismiss, he has to assume that everything we’re saying is true. And I think it was just done to delay us. And I can’t imagine a judge giving a correct decision here that wouldn’t at least allow us to have a factual hearing. And the reason we came to you and some of the other state legislators is because I believe that we’re being denied our right to a hearing by the courts. But then again, you do have responsibilities equal if not superior to the courts to oversee these elections.
Rudy Giuliani: (03:20:03)
So we’ve made similar requests to the legislature in Michigan, Arizona, and Georgia. And we’re very hopeful that next week we’ll get to present to them as we also press for a hearing in court. I think it is really important for the American people to see these are not my claims or Jenna’s claims or President Trump’s claims. And many of these people came to us because they’re so upset. These are the complaints of the American people. And I’m more than happy to make available to you an outline of the other affidavits. It would have been impossible to produce all the witnesses, but I can show you that for each one of these witnesses, there are anywhere from 15 to 50 other witnesses who saw the same thing.
Rudy Giuliani: (03:20:54)
I have a list just in Pittsburgh alone of 27. I have a list in Philadelphia of 32. I have a list of about 40 witnesses on cure. Even though the numbers aren’t ascertainable, there’s still a lot. None of the things we’re saying to you are just arguments. This this evidence. I keep explaining to people, affidavits are the best evidence you can have until somebody actually gets into court and swears. And our evidence is under oath. Their claims are just claims.
Senator David Argall: (03:21:30)
We have one additional question of online from Representative-
Rep. Daryl Metcalfe: (03:21:33)
Thank you.
Rudy Giuliani: (03:21:33)
Thank you, sir.
Senator David Argall: (03:21:34)
Thank you, Daryl. From Representative Keefer.
Rep. Daryl Metcalfe: (03:21:36)
Thank you, Senator Argall. Thank you, Senator Argall. Thank you, Mayor Giuliani.
Senator David Argall: (03:21:43)
Representative Dawn Keefer.
Rudy Giuliani: (03:21:43)
Thank you.
Rep. Dawn Keefer: (03:21:45)
Thank you. Thank you, Senator Mastriano for hosting this, and thank you mayor, for all of the testimony that you have brought forward. Two questions I have. First is regarding testimony that we received on the pink highlighted ballots that were scanned through and submitted, and then they were able to be re, I guess, re-documented, and then scanned through again. Is there any action being taken on that, given the number of ballots that we’re talking about there?
Rudy Giuliani: (03:22:13)
Do you have the answer to that?
Speaker 14: (03:22:19)
I don’t. That’s a state issue.
Rudy Giuliani: (03:22:20)
I’m not know the answer to that, but I’ll get it for you.
Rep. Dawn Keefer: (03:22:24)
Okay.
Rudy Giuliani: (03:22:24)
That’s not something we [crosstalk 00:10:25]-
Rep. Dawn Keefer: (03:22:30)
Okay. That would be great to know-
Rudy Giuliani: (03:22:30)
We will find out. That’s a very good point and-
Rep. Dawn Keefer: (03:22:30)
Because I know we saw a lot of that, either speculation [inaudible 03:22:32] that people were just blindly filling out these ballots, but there is supposed to be a process. And it was my understanding that that process was supposed to be videoed or live streamed and witnessed by majority and minority inspector when you are recreating a ballot that is not able to be scanned. So that would be good to know.
Rep. Dawn Keefer: (03:22:52)
And the second thing is, Attorney Ellis. You had made a comment regarding what our constitutional ability is. And so while we understand what the Constitution does say as far as us taking responsibility and we could intervene in certain sectors, while the Constitution gave us that ability, it’s been explained to us by attorneys that we took that power that was given to us as General Assembly members and we put it into statute, and that statute says how we appoint our electors and how we certify the elections. It’s all spelled out in our Pennsylvania Code based upon the power that we have from the Constitution.
Rep. Dawn Keefer: (03:23:38)
You have a differing legal opinion of that.
Jenna Ellis: (03:23:41)
Yes, I do. And I think that our collective, and with my co-counsel here, you can take that power back at any time. And when you have the delegation of authority, the legislature can take that back. And you also have an instance here that the law was completely ignored. So even though you have a manner in which your electors are generally selected in Pennsylvania, and that’s worked for the past presidential elections since those statutes were authorized and gone through the legislature, this is an election that has been corrupted. And so you can’t go through that method, and those laws were violated, and the General Assembly here, or the legislature, is the authorized entity in the Constitution that selects the manner.
Jenna Ellis: (03:24:29)
You can take that power back at any time. And so when the laws were ignored and that’s where we have and why we have all of this evidence to bring to you, is to show you each of these different types of ways that election officials have violated the manner in which you, as the General Assembly have prescribed in the law, how you select your electors. Because they violated that law, you have the constitutional authority to craft a remedy. You can take that power back at any time. You don’t need a court to tell you that. So, absolutely, and you can look at that. You can look at the federal law for elections. This is your constitutional prerogative. State legislature in the context of Article Two, section 1.2, means state legislature, period.
Rudy Giuliani: (03:25:14)
You also delegated it to the very people whose conduct is in question, and you’re asking them to investigate themselves. So given the fact that this is your sole constitutional right and authority, you can always assume constitutional authority that you’ve delegated back. You also have a rational basis for doing it because you’re asking them, I mean, this was done, one might argue, or at least that’s one of the interpretations of the fact, deliberately by the Governor, by the Elections Commissioner, who issued absurd opinions telling people to violate the law. And then you’re going to ask them to make a decision on the election that allegedly they corrupted. It would seem to me, you have a perfect right to take that back and make that decision to no vote.
Senator David Argall: (03:26:12)
Thank you. Thank you very much. I want to thank everyone for your patience, the public, our many witnesses, our senators and representatives. As you’ve seen today, this COVID pandemic has made holding a public hearing a little bit more challenging than usual, but we have still received an enormous amount of information today, information that we will share with every one of our members. There is no doubt in my mind that we will pass legislation in the House and the Senate based on what we have learned today. I also want to thank a Senator Mastriano. Doug and his staff have been enormously helpful today. It really goes without saying we would not be here without Doug Mastriano, and so I’m going to give him the last word because I know even if I didn’t, he’ll take it anyway, and he deserves to have it. Doug.
Senator Doug Mastriano: (03:27:23)
I love you, Senator Argall. Thank you for being willing to use your committee to do this hearing. So we pulled the trigger yesterday morning around 9:00 AM, and look what happened here. And as Jesus said, “You shall know the truth and the truth shall set you free.” Guess what? Guess what? The truth’s out there. Media, you should have found these witnesses. You need to do your job. Now you see them out here. Do your job. You’re essential to this Republic, and without you doing your job instead of being partisan hacks. This Republic can’t stand. So now you got some information. You got some witnesses with courage. These people are heroes to me. As a retired Army Colonel, I look at them and I say, you guys are outstanding. Thanks for being willing to step up.
Senator Doug Mastriano: (03:28:07)
Because I am cognizant that the so-called tolerant people on the left will not show much love or tolerance to people they disagree with. And so thank you for standing, because you are warriors being able to stand for and save this Republic. So thank you for going forward here. I mean, indeed, I started as off quoting for John Adams. We are in Adams County. You know, facts are stubborn things. There’s a lot of hard facts out there. And guess what? As history and change in 1863 in Gettysburg back then, it’s July first, second or third. So on this day, history is changing for our country and state back at Gettysburg once again.
Senator Doug Mastriano: (03:28:49)
I mean, I don’t even know how this happened in America. We could send, 50 years ago, men to the moon, but we can’t have a safe, secure election in Philadelphia in Pennsylvania. What’s going on here? It’s got to be by design because we have the technology. We have stealth aircraft that are the envy of the world, but we can’t run an election better than Afghanistan? Congressman Perry, you and were in Afghanistan, and I don’t know how in the heck Afghanistan has more secure, safe elections than Pennsylvania does. I mean, beam me up Scotty. There’s no sign of intelligent life anywhere. Really. We move heaven and earth with American dollars to secure elections in Iraq and Afghanistan and elsewhere. We can’t do it in our own state? It’s by design. There’s people in Pennsylvania, not interested in safe, secure elections, and we have to correct this. There’s two things that need to happen.
Senator Doug Mastriano: (03:29:34)
First off. We need to make sure that the real winner is sent forth from this presidential election. And number two, we need to fix this so this doesn’t happen ever again. You know, I can’t believe, I can’t believe we’re having this conversation here in Pennsylvania. You know, this is the kind of stuff I hear about that goes on in Belarus under Lukashenko or in Russia under Putin, or under Saddam Hussein’s Iraq. Really? Here in our state? I mean, this is disgusting to me. You know, I watched, when I was doing strategy in Russia, Putin’s people stuffing ballot boxes, the same thing that happened in Pennsylvania. When I saw that 15, 20 years ago was like, “Well, thank God we’re not like that.” What happened? What happened?
Speaker 15: (03:30:22)
We won’t let it stand.
Senator Doug Mastriano: (03:30:23)
And we’re not going to let it stand. You know, you have to forgive people because we are a constitutional Republic, so I’ll forgive those that say democracy, but I saw one man had a sign in Philadelphia and the irony wasn’t lost on me. Democracy dies in Philadelphia. Can you imagine the irony of that? You know where the light of liberty was lit in 1776, it transformed this world where in this very same state, a new birth of freedom. And then of course let’s not forget 2001, Todd Beamer, let’s roll. You know what? It’s our time to roll. This is no time. You know, Democracy can die in darkness, in dark rooms with no transparency and accountability. We’re shining light on his darkness here. We’re going to take our state back. We’re not standing aside in this hour. You know, as Representative Metcalfe rightly said, in 1994, there was so much shenanigans and cheating in the Marks v. Stinson case that a federal district judge throughout the election results, took a Senator out of Harrisburg, state Senator, and put the Republican in because the election results were of course so corrupted. So we do have a precedent here, federal courts, and I hope you take a hard look at that here.
Senator Doug Mastriano: (03:31:34)
Has our election results in Pennsylvania at the presidential and other levels been so corrupted that the results have to be thrown out? That’s going to be a case you’re going to have to make before the Supreme Court there, but it sure sounds like something stinks in Denmark or in Philadelphia. I don’t know. You know, one vote, one person, one legal per person, and any cheating goes in there, disenfranchises an American. I can’t believe this is happening here and we’re not going to let it stand. I don’t know why it’s so hard. You know, I know it’s hard because this has been going on here for a lot longer than we imagined. But in Galatians 6:9, we’re told by St. Paul, “Grow not weary doing good because in due time you’ll reap your harvest.” We’re going to reap our harvest.
Senator Doug Mastriano: (03:32:21)
The time for the dithering and deliberation is over. It’s time for decisive action. We have to protect our Commonwealth and our nation. The eyes of the world are upon us and let’s turn the Commonwealth of Pennsylvania from a laughing stock to the pride of the world once again, as we have always been and need to be again. This is our day. This is our hour. This is our time. So yes, Mr. Franklin, a Republic if you can keep it. We’re going to keep it. Can you keep it? Can you? Will you? We’re going to keep the Republic. Thank you and God bless you all.
Senator David Argall: (03:33:03)
Thank you all. We stand adjourned.
Senator David Argall: (03:33:04)
(silence)
Pennsylvania Senate Republican Lawmaker Hearing Transcript on 2020 Election
Anomic Age Podcast Interview re: Perverted Truth Exposed
Anomic Age Podcast interview with Kay Kiser, the author of “Perverted Truth Exposed: How Progressive Philosophy Has Corrupted Science”

Kay Kiser is with us today to discuss the proliferation of the progressive philosophy throughout academia, her books, Darwin, and more.
Kay is a retired chemist with over 30 years of experience in industrial research and development. Professional roles include microbiologist, teacher, technical writer, inventor, lab manager/designer, and volunteer crisis counselor. She hold 5 patents, wrote/taught many technical manuals and procedures, and coauthored a chapter in an American Chemical Society book, “Radiation Curing of Polymeric Materials”. Mrs. Kiser is a lifelong researcher into all things related to science and history. She hold a Lifetime Achievement Award from Marquis Who’s Who, which has been featured in the Wall Street Journal, Nov. 26, 2018 and other publications. Kay is a conservative Christian, skeptic, iconoclast and seeker of truth.
She has published two non-fiction books in her Modern Mythology
Series, pointing out the myths that most of us have assumed to be true.
Her first book, “Perverted Truth Exposed: How Progressive Philosophy Has Corrupted Science”, presents logical arguments questioning long-standing scientific assumptions and world views in which progressive philosophy has been disguised as science in a nested series of theories. Examination of the history, philosophy and political connections reveals the flawed logic of many scientific theories. Academia has become a closed priesthood, not an open forum for truth. Adherence to the party line determines what research is done, funded and published.
Kay’s second book, “Saving Africa from Lies that Kill: How Myths about the Environment and Overpopulation are Destroying Third World Countries” is an Award-Winning Finalist in the Social Change category of the 2019 International Book Awards. It exposes long-standing crimes by international organizations that keep developing nations poor, isolated, ignorant, sick and, most of all, controlled, all based on the myths that overpopulation has caused environmental harm, and assumed genetic inferiority of impoverished cultures. Poverty, not overpopulation, causes environmental harm.
Return to the God Hypothesis
The Intellectual and Moral Decline in Academic Research — The Wentworth Report
The Intellectual and Moral Decline in Academic Research, by Edward Archer. My experiences at four research universities and as a National Institutes of Health (NIH) research fellow taught me that the relentless pursuit of taxpayer funding has eliminated curiosity, basic competence, and scientific integrity in many fields. Yet, more importantly, training in “science” is now […]
via The Intellectual and Moral Decline in Academic Research — The Wentworth Report
Peer Review Is Bunk — Big Picture News, Informed Analysis
A report I wrote for the Global Warming Policy Foundation was released today. It explains that peer-reviewed research is as likely to be wrong as right. Basing public policy on findings that haven’t yet been reproduced is nuts.
via Peer Review Is Bunk — Big Picture News, Informed Analysis
Balanced, Static Nature: The Grand Illusion — Big Picture News, Informed Analysis
Change is natural and normal. Let us eat, drink, and be merry.
via Balanced, Static Nature: The Grand Illusion — Big Picture News, Informed Analysis