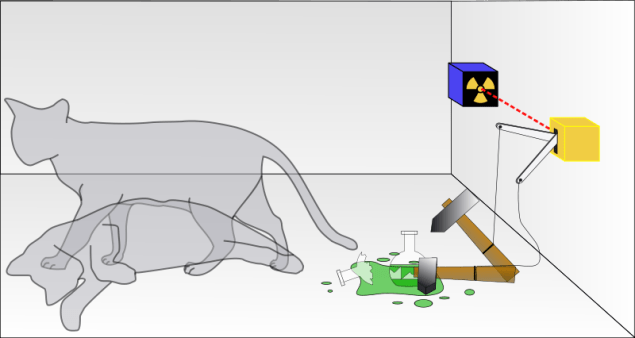
Quantum Theory – Recent Hype and Magical Thinking about saving Schrodinger’s Cat prompted me to bring back and revise this post that lays out logical and historical facts about Quantum Theory interpretations and implications in plain language for the average non-scientist.
Quantum Mechanics or Quantum Theory, which is based on complex mathematics, tries to describe and explain the odd behavior of particles and forces in the atomic and subatomic realm. In this theory, things don’t happen in a smooth (analog) manner but in a punctuated (digital) manner.
The Basics: Electrons move around the nucleus of atoms at high speeds so that their exact location at any one moment is not known precisely without measurement. The likelihood of finding a given electron at a particular place in its orbital is described by a probability, thus defining the electron “cloud” or “shell.” An electron jumps from one allowed orbital to another by absorbing energy (a photon) at a specific energy level (wavelength).
The absorbed photon at a specific energy level is called a quantum, thus quantum theory. The electron will also fall from this “excited” state back to its more stable “ground” state orbital by emitting a quantum of energy. Electrons exist or move between one allowed energy state (orbital) and another based on discrete quanta of energy that they absorb, emit or carry. Each element has unique orbital energies so that light interacting with an atom shows absorption and emission lines at specific wavelengths that can be used to identify the element.
Wave-Particle Duality:
In Quantum Theory, subatomic particles are described as both particles and waves simultaneously. This is referred to as wave-particle duality. All types of energy, including subatomic binding forces, are also defined as both particles and waves, so that matter and energy are treated as if they are the same thing. Both subatomic particles and photons sometimes act like waves and sometimes like particles, depending on how they are tested or detected. Two experiments are noted as evidence: the double slit interference patterns and the photoelectric effect in which electrons are emitted when light is shined on a metal surface. The first experiment seems to show that particles are waves. Einstein assumed the second experiment proved that energy waves were really made of particles that he called photons.
The double slit experiment is said to demonstrate the wave nature of particles and photons. The photoelectric effect is said to demonstrate the particle nature of particles and photons. Wave-particle duality rests on the assumption that single photons or particles are being measured. Since all detectors have threshold sensitivities below which nothing is detected, it could mean that multiple, not single, photons or particles are really being tested[1]. This would explain the interference patterns seen when either photons or electrons are tested in the double slit experiment that makes it appear that a single particle went through both slits at once. Photoelectric experiments may also be misinterpreted. It is possible that absorbed energy, not photon particles, causes emission of loosely held electrons on the metal surface. Granted, this is speculation at this time, but calls for more study.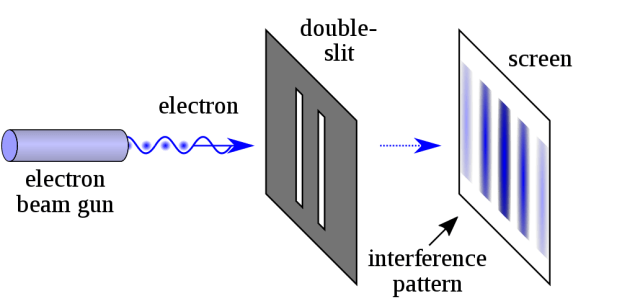
Attribution: Double Slit Experiment – from Wikipedia by NekoJaNekoJa and Johannes Kaliauer 2017 under Creative Commons Attribution-Share Alike 4.0 International license.
Copenhagen Interpretation:
In the widely accepted Copenhagen interpretation of quantum mechanics, a particle is said to not have a fixed state but exist in a smeared out multiplicity of states at once until a measurement is taken when it “collapses” into one state. Magical Thinking Alert! The observer (or detector) becomes a part of the quantum system. This is the principle of superposition. Because an electron can be found in any of the probability-allowed “shell” locations, this interpretation assumes that the electron really is at all the locations or states at once and only assumes a fixed state when measured. This assumption extends to all of the characteristics of the electron such as position, spin or momentum. This assumption also extends to all other subatomic particles and photons (energy particles).
The Copenhagen interpretation of Quantum Theory also says the electron exists in one or the other allowed orbital level but does not exist anywhere between. When a quantum of energy is absorbed the electron is said to pop out of existence in the original shell and simultaneously pop into existence in the new shell. But, since the electron shell defines a probability, and most of the time the electron exists in one of these shells, the probability of finding it anywhere between is statistically infinitesimal. It is said not to exist there, and it is thus called “forbidden.” Is it only an extremely small probability or are we talking about its actual existence? The Copenhagen interpretation of Quantum Theory says it is the latter. Other interpretations of Quantum Theory differ as to what actually happens.
Uncertainty Principle: Ontology or Epistomology?
In trying to measure these discrete orbitals and their electron locations and momenta, it became apparent that measurement of any kind disturbed the system so that only one of two coupled parameters could be determined at any one time, e.g. position and momentum (or speed). This led to the Heisenberg Uncertainty Principle, which states that it is impossible to know both the position and the momentum of any one subatomic particle at the same time. The system is disturbed by measurement because measuring subatomic particle parameters is like administering eye drops with a fire hose. Because the subatomic particles are so small compared to any means of measuring their parameters, what is measured is in a disturbed condition.
The Heisenberg Uncertainty Principle was meant to be a statement of experimental limitations, not that location and momentum (or other coupled parameters) did not exist in a fixed state at the same time. However, Niels Bohr, its originator, and other Copenhagen interpretation proponents interpreted it that way, assuming that atomic particles were never in a fixed state until measured, and that uncertainty is a fundamental characteristic of subatomic particles, not just an experimental limitation. Thus they have substituted ontology (being) for epistemology (ability to know). Heisenberg never accepted the principle of superposition or non-locality claimed in the Copenhagen interpretation.
Superposition:
Edwin Schrodinger provided the mathematical equations for the behavior of electromagnetic waves that are used in quantum mechanics[2]. These probabilistic differential wave equations are linear (first order), that is, they can be plotted as straight lines on a graph. Superposition is a concept in mathematics stating that in linear equations all of the contributing factors must add up to the net effect of each factor individually. Since Schrodinger’s equations for waves are linear, it is assumed that their application to subatomic particles is also linear. From there it is a leap of faith to assume that particles don’t just have the capability of being in different states, but that they are simultaneously in all possible states at once. Instead of just being a mathematical concept, superposition now was applied directly to subatomic particles in a real physical sense.
However, Schrodinger, who created the wave equations, did not agree with this Copenhagen interpretation of quantum mechanics. He came up with an example within everyone’s sphere of experience that illustrated the absurdity of their assumed superposition. This was the famous Schrodinger’s Cat thought experiment. He set up the experiment so that a cat in a closed box could be either alive or dead, depending on whether a radioactive particle spontaneously decayed setting off a mechanism that released a deadly poison gas. In this thought experiment, using the Copenhagen interpretation of superposition, since we don’t know what state the cat is in until the box is opened, the cat is both dead and alive until it is opened at which time the cat becomes either dead or alive. The act of observing somehow must cause the cat to assume either a dead or alive state. In all other realms, this would be called Magical Thinking. It was meant to point out the weakness or absurdity of superposition, but it has been misused to illustrate the opposite through convoluted “reasoning” to make it fit the Copenhagen or similar interpretations. Recent efforts are no different, regardless of hype.
Diagram of Schrodinger’s Cat Thought Experiment Attribution: From Wikipedia by Dhatfield, 2008 under Creative Commons Attribution-Share Alike 3.0 Unported license
Communication at a distance:
The idea of instantaneous communication and action at a distance is a consequence of this assumed superposition where particles do not assume a fixed state until observed. By Pauli’s Exclusion Principle, no two electrons in the same orbital can be in the same quantum state. Each must differ in some way, for example they must have opposite spins. The two particles are said to be entangled since each must be in the opposite state to the other. If one of the electrons is emitted and travels relatively far away, when one of the electrons is measured (observed), it collapses into a fixed state and simultaneously the other one collapses into the opposite state that can be confirmed when it is measured. This implies speed of communication faster than the speed of light, the assumed upper limit of speed[3].
Einstein thought that quantum action at a distance was an illusion based on the assumption of superposition, aka non-locality. If particles are assumed to have fixed states, although unknown to an observer, the action at a distance is no mystery. It only implies that entangled states, e.g. opposite spins, persist after separation. When one of the particles is measured, you automatically know the state of the other since they must be the opposite of each other, whether together of separated. Einstein spent the latter part of his career trying to prove this, but was ignored and sidelined because he did not go along with the accepted theory. His years-long arguments with Niels Bohr are historically noteworthy.
Other Interpretations:
There are more than a dozen other interpretations of quantum physics. The most popular, among a long list, (see table following), are the Copenhagen interpretation and its variants, the Many Worlds interpretation and the Ensemble interpretation. Variants of the Copenhagen interpretation involve either the observer or the cat (as observer and participant) as being parts of the quantum system. Another, the Many Worlds interpretation is even more speculative. In this scenario, each time a subatomic particle collapses and “chooses” a fixed state, reality splits in two and both possible realities still exist, but in different undetectable dimensions. Think of this as a time series of pictures or a strip of movie film. At the decision point, the one series becomes two, and at the next decision point, becomes four, etc. ad infinitum.
The Ensemble interpretation states that Quantum Mechanics can only be applied to statistically significant numbers of particles, not to individual particles. Since the wave equations describe probabilities, it would be meaningless to apply probabilities or statistics to single particles. This is the interpretation favored by Einstein but is discounted by leading QM physicists. Similar realistic interpretations such as those proposed by de Broglie-Bohm and science philosopher Karl Popper assume real particles with real positions and real wave functions that do not need to “collapse” upon measurement. I tend to prefer these theories because of their realism.
“The attempt to conceive the quantum-theoretical description as the complete description of the individual systems leads to unnatural theoretical interpretations, which become immediately unnecessary if one accepts the interpretation that the description refers to ensembles of systems and not to individual systems.”
—A. Einstein in Albert Einstein: Philosopher-Scientist
Is the universe really indeterminate?
As a consequence of the probabilistic view, by the Copenhagen Interpretation of the subatomic world, Quantum Theory leads to a conclusion that events are not deterministic, but rather are indeterminate; that they just happen without actual connections between cause and effect. If deterministic, then events in the past must predict future events as causal antecedents. In the macro or “real” world, everything has a cause or causes, whether known or not. Determinism is the accepted view or apparent state of the real universe because, knowing the mass, position and the momentum of a (larger) body, plus all of the influences on it and the mathematical equations governing its movement, one can (in theory) calculate its position and speed at any other time in the future or the past. This is the basis of celestial mechanics by which planets, etc. are tracked.
The question is: since we don’t know for sure what the outcome according to QM will be, is it really indeterminate or are there certain things we don’t or can’t know about the system that only makes it look indeterminate? If it were possible to know all of the parameters and influences without disturbing the system could we, with certainty, predict outcomes? According to the Copenhagen interpretation of quantum theory, the universe is really indeterminate at the atomic level and only LOOKS determinant at the macroscopic level. This eliminates the infinite series of cause and effect, and therefore the question of a first cause that started it all and, to some, the uncomfortable possibility of God as a necessary being.
[1] See also Andrew Ancel Gray at http://modelofreality.org/cgi-bin/iet.cgi
[2] Side note: These equations assume massless particles and waves. Since real particles have mass, particle physicists assume there is a particle that gives all other particles mass. The Higgs boson is the assumed particle that creates mass when a particle is in a Higgs field.
[3] It should be noted that many thought experiments and most actual experiments have been done using light, not subatomic particles. The results of these actual experiments depend on your interpretation of Quantum Theory. See other interpretations that follow.
Major QM Interpretations (click to follow link)
**********************************************
 Want to know more about this and other Modern Myths including climate change, evolution, origin of life, Big Bang cosmology or quantum physics? See related posts on this website, www.realscienceblog.com, or buy the book Perverted Truth Exposed: How Progressive Philosophy Has Corrupted Science on line at Amazon, Books-a-Million or Barnes & Noble .
Want to know more about this and other Modern Myths including climate change, evolution, origin of life, Big Bang cosmology or quantum physics? See related posts on this website, www.realscienceblog.com, or buy the book Perverted Truth Exposed: How Progressive Philosophy Has Corrupted Science on line at Amazon, Books-a-Million or Barnes & Noble .
From the back cover:
In Perverted Truth Exposed, Kay Kiser exposes areas of science that have been corrupted by progressive and atheist philosophies disguised as science, including the theories of evolution, origin of life, cosmology and quantum physics. The Climate change debate presents a modern example of how the perversion of science is politically imposed to support an anti-God, anti-human progress agenda of Marxist control and power while silencing opposition through intimidation. Kiser also answers:
- Did Darwin really steal his theory of evolution from Alfred Wallace? Why did Wallace later abandon the theory as not having sufficient evidence?
- If Hubble discovered the expanding universe leading to the Big Bang Theory, why did he continually try to convince others that their conclusion was wrong?
- Is man made carbon dioxide causing global warming or is it a trailing indicator of climate change in a system dominated by solar cycles, cloud cover and ocean currents?